Search any question & find its solution
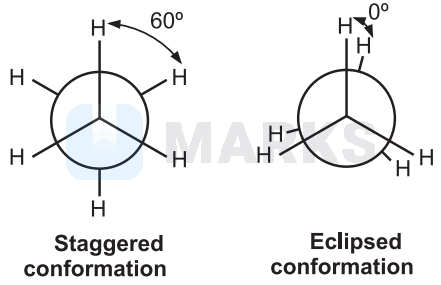
In staggered conformation arrangement, the hydrogens of the two carbon atoms are staggered with respect to one another. As a result, they are at maximum distance apart and have minimum repulsion between the hydrogen atoms attached to the two carbon atoms.
In eclipsed conformation, the hydrogens of one carbon atom are directly behind those of the other. Consequently, the repulsion in between these hydrogen atoms is maximum. Therefore, staggered conformation of ethane is more stable than eclipsed conformation.
Eclipsed conformation is the least stable conformation of ethane.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.