Search any question & find its solution
Question:
Answered & Verified by Expert
The increasing order of basicity for the following intermediates is (from weak to strong)
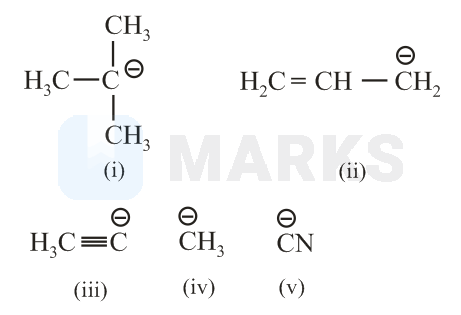
Options:

Solution:
1939 Upvotes
Verified Answer
The correct answer is:
Basicity is inversely proportional to electronegativity.
Higher the electron affinity or core charge of an atom, the less likely it is to donate its electrons to a proton.
Greater the delocalization of electrons that could potentially donate to a proton, the less able they are to donate.
Greater the electron-withdrawing effects in another part of a molecule, the less likely the electrons on a particular atom are to donate.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.