Search any question & find its solution
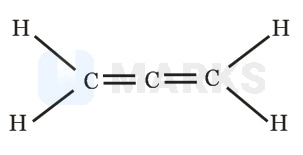
Which of the following statements correctly gives answers for all 3 parts:
(a) Is the molecule planar?
(b) Does 1, 3 - dichloro propadiene show geometrical isomerism?
(c) Is the molecule 1, 3 - dichloro propadiene polar?
(ii) No geometrical isomerism
(iii) Polar
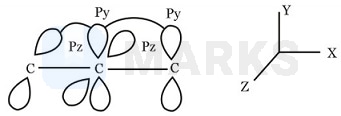
Hybridization at the terminal carbons are both . The two p - orbitals involved in hybridization are and at left terminal and and at right terminals. Therefore, the three hybrid orbitals at left terminal carbon are in plane, while the same at right terminal carbon are in
plane. As a result, the two triangular planes at terminals are at right angles as shown below:

The two planes at terminals are perpendicular to each other. ( - bonds are also perpendicular to each other.)
Hence, (a) Molecule is non - planar. (b) It doesn't show geometrical isomerism because two H and chlorine are in different planes. (c) polar.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.