Search any question & find its solution
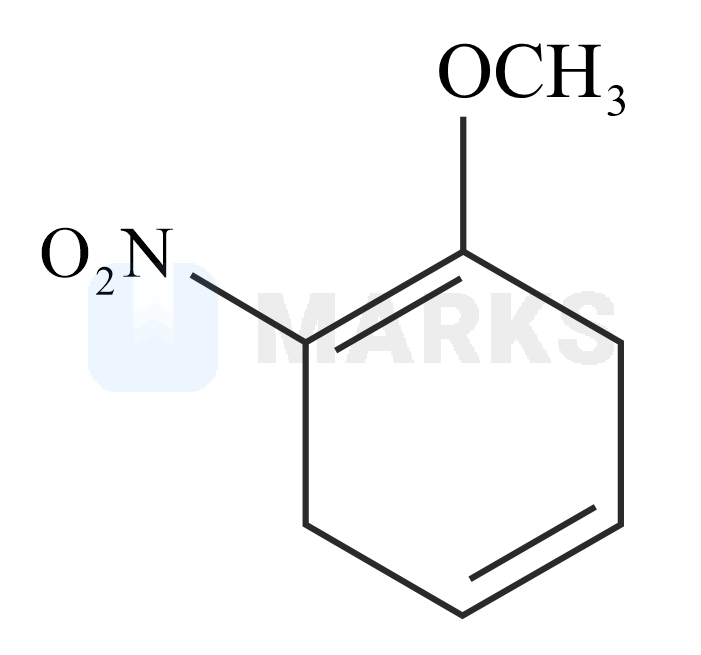
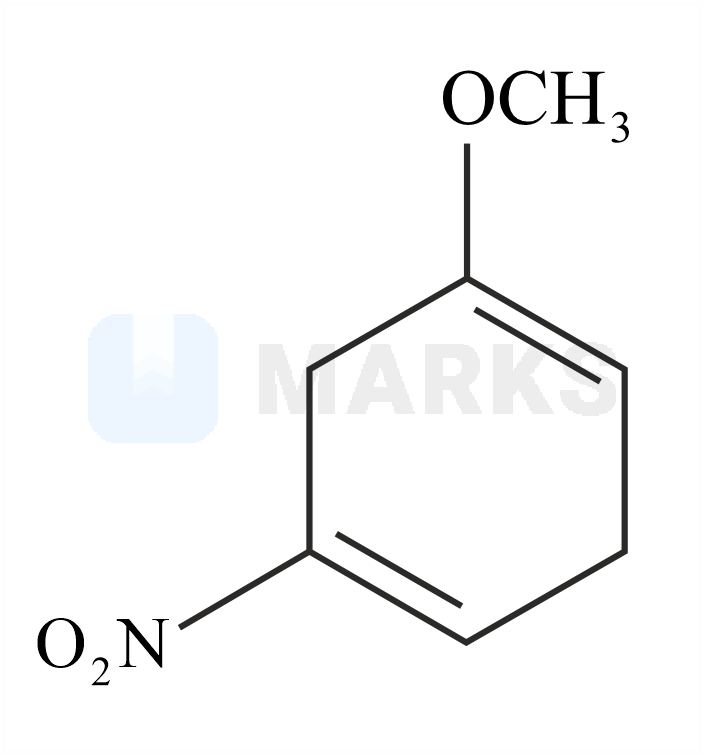
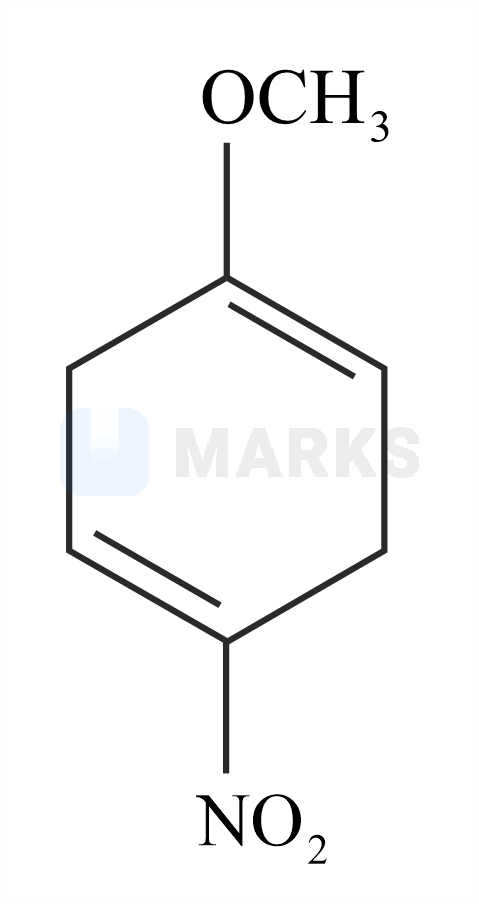
The major product formed in the given reaction is:
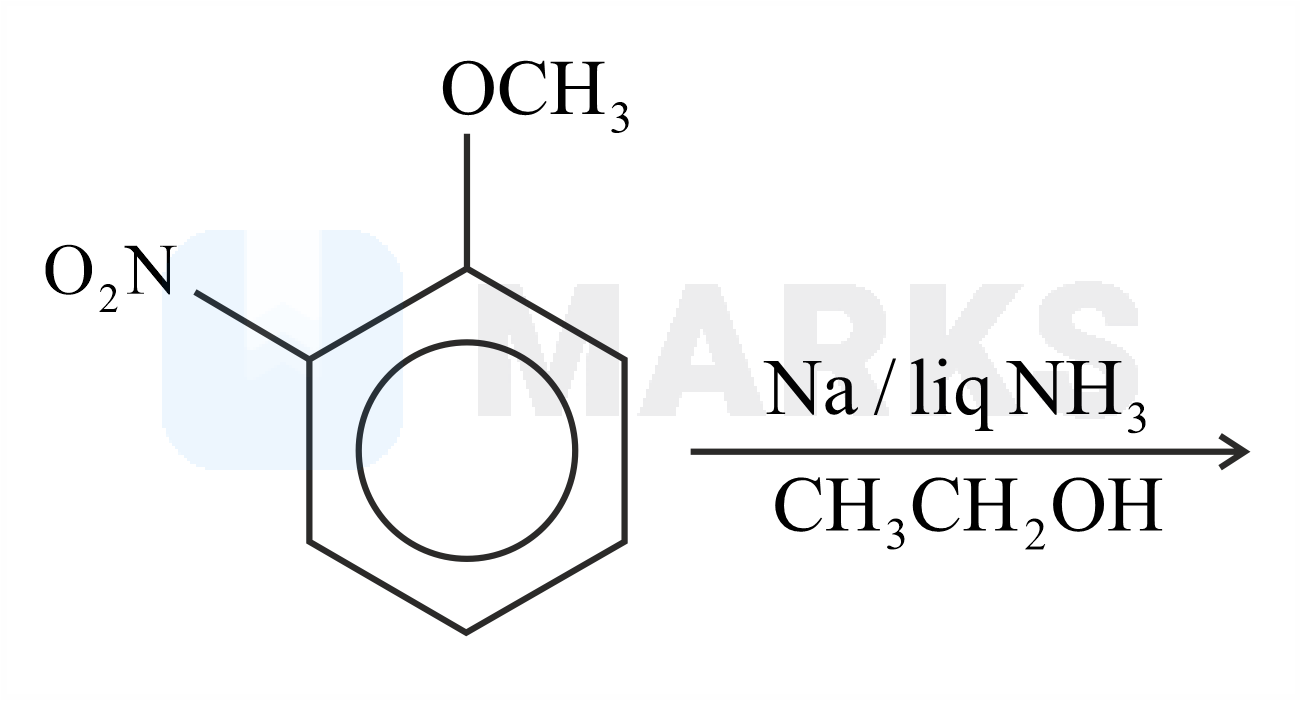
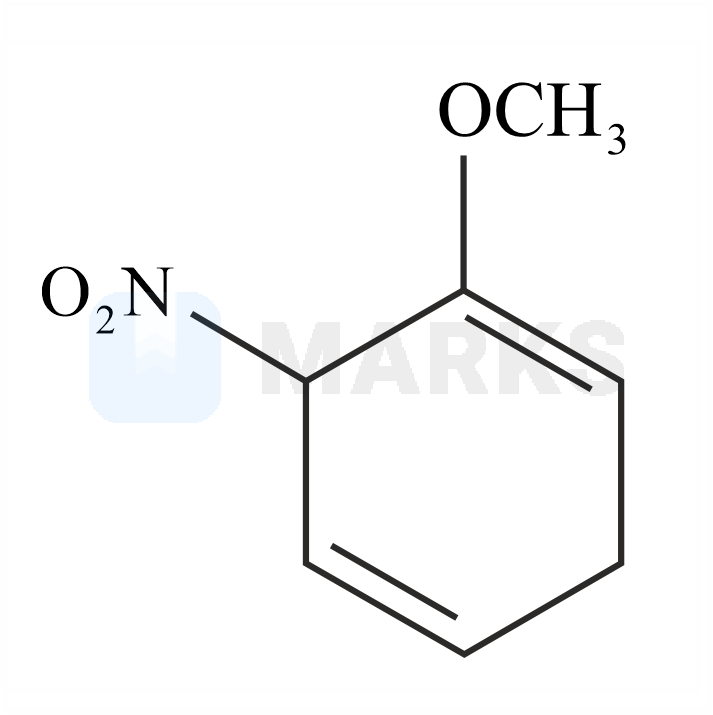
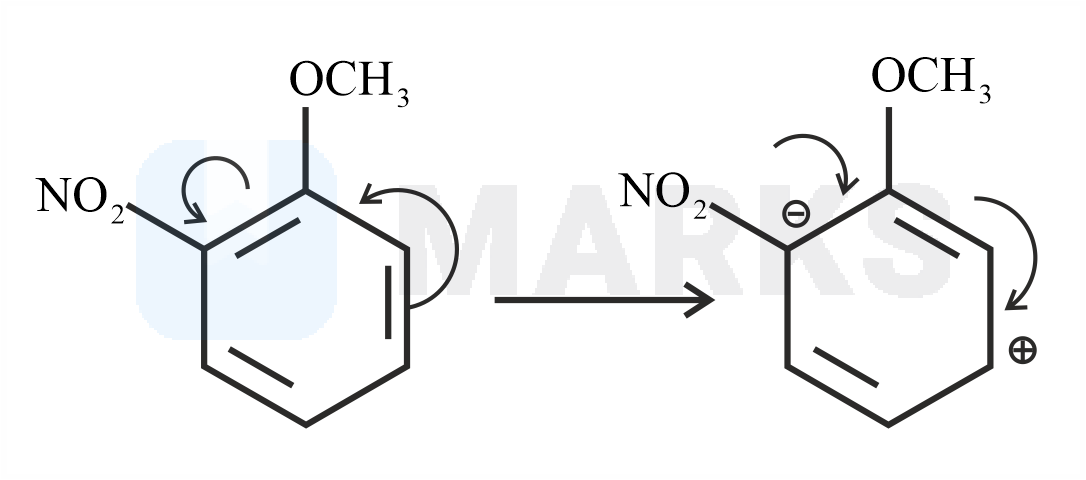
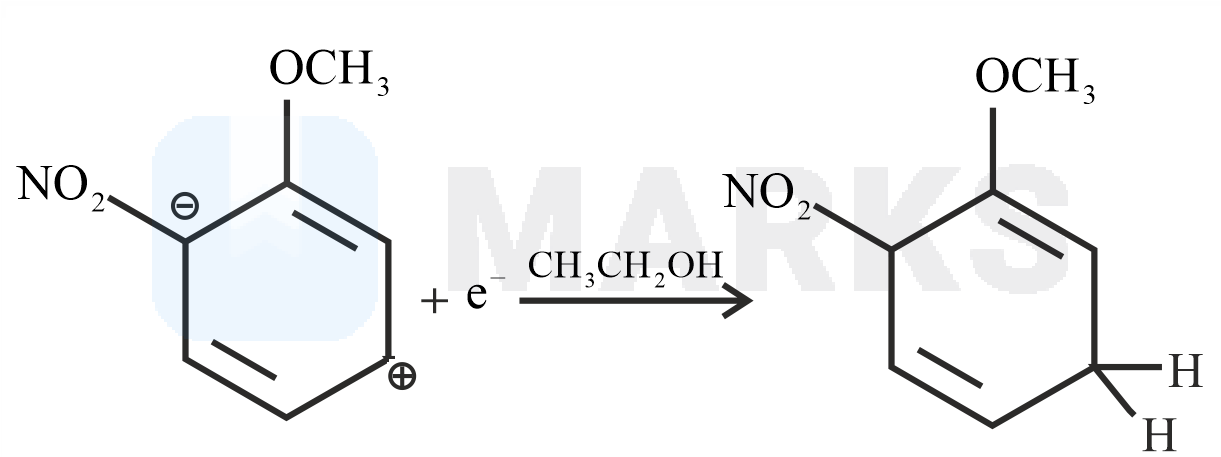
The Birch reduction is an organic reaction where aromatic compounds which have a benzenoid ring are converted into 1,4-cyclohexadiene which have two hydrogen atoms attached at opposite ends of the molecule. The solvated electrons (the free electrons in the solution of sodium in liquid ammonia which are responsible for the intense blue colour) add to the aromatic ring, giving a radical anion. This radical anion is supplied with a proton by the alcohol. The alcohol also supplies another proton to the penultimate carbanion. Now, with the alcohol present, the products 1,4– cyclohexadiene and an alkoxide ion are formed.
The reaction with nitro anisole is given below.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.