Search any question & find its solution
Question:
Answered & Verified by Expert
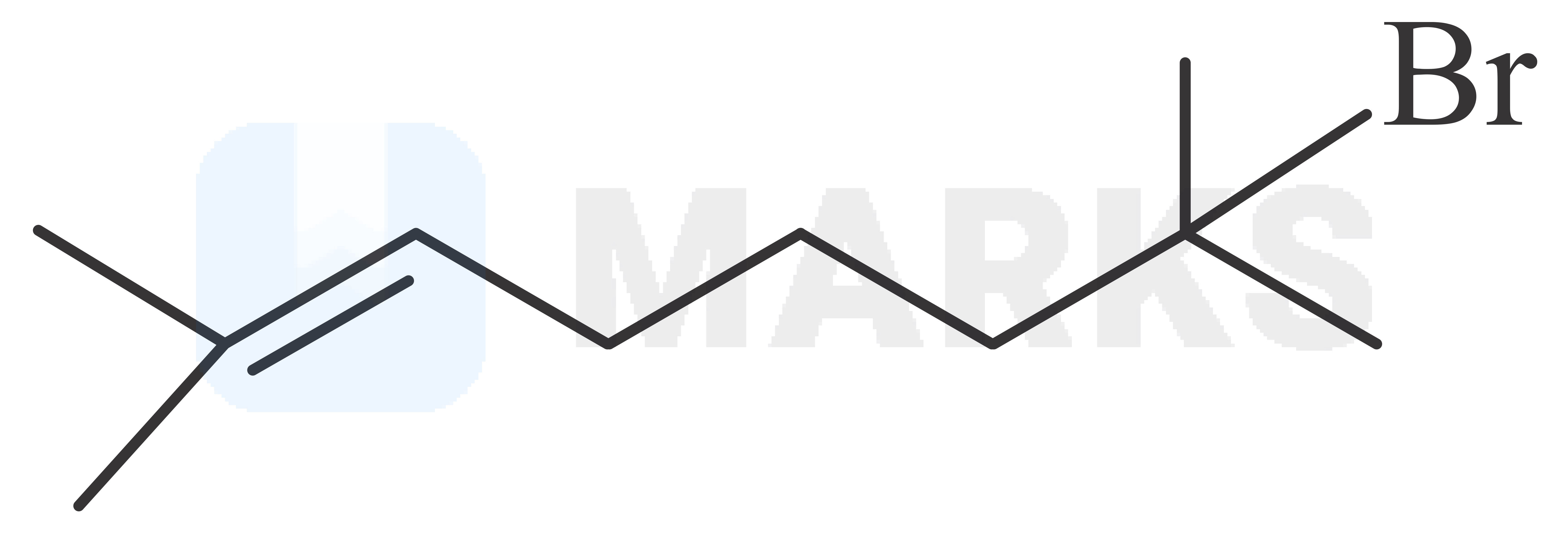
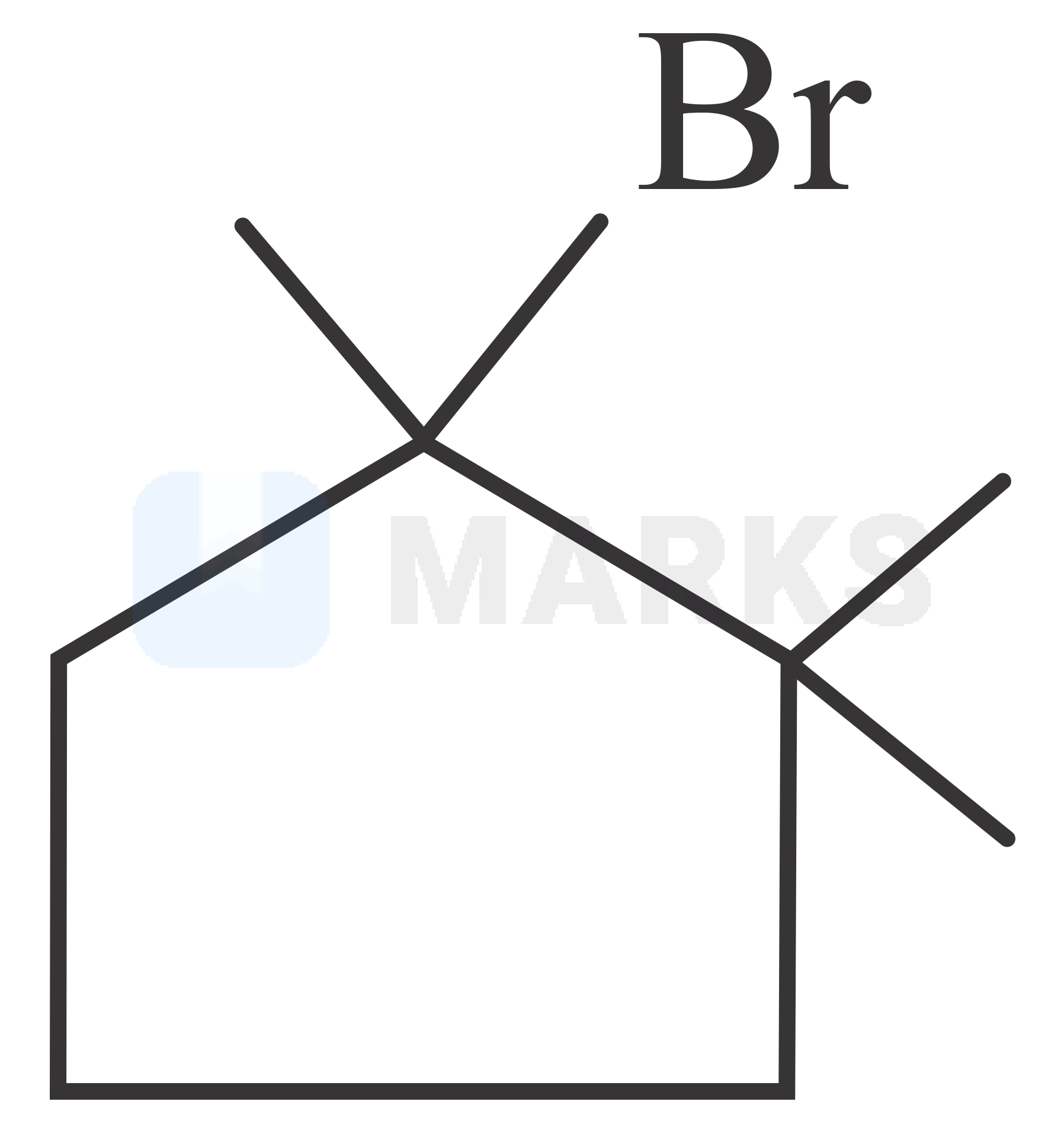
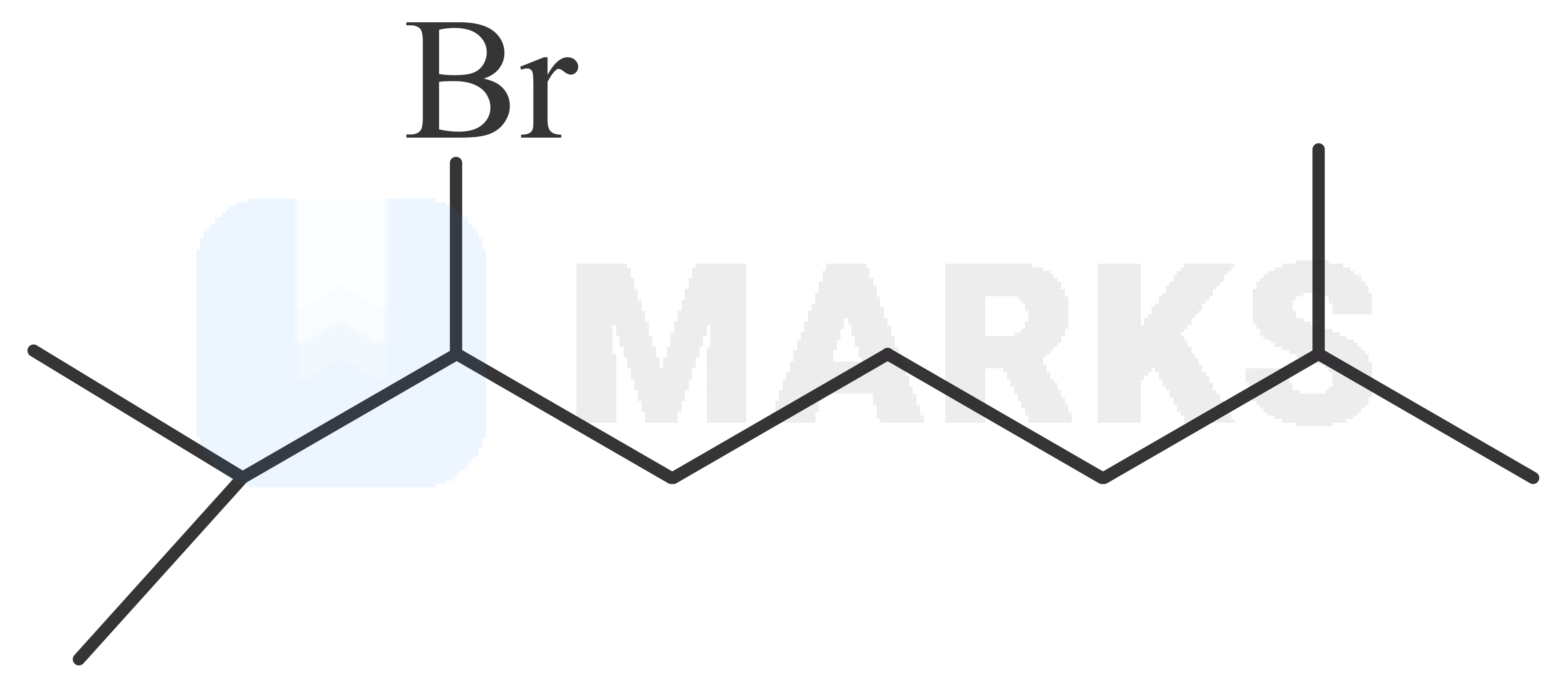
The major product in the given reaction is
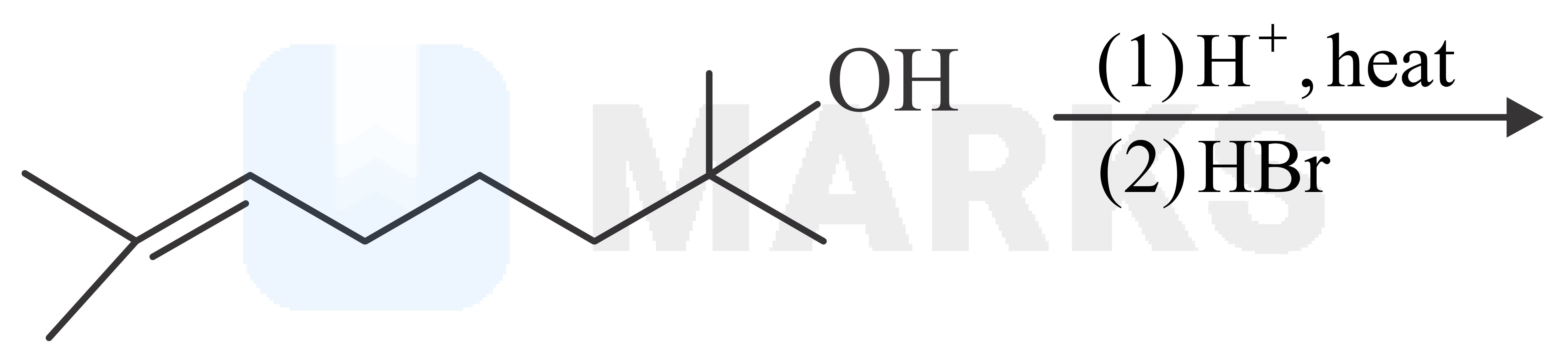
Solution:
1326 Upvotes
Verified Answer
The correct answer is:
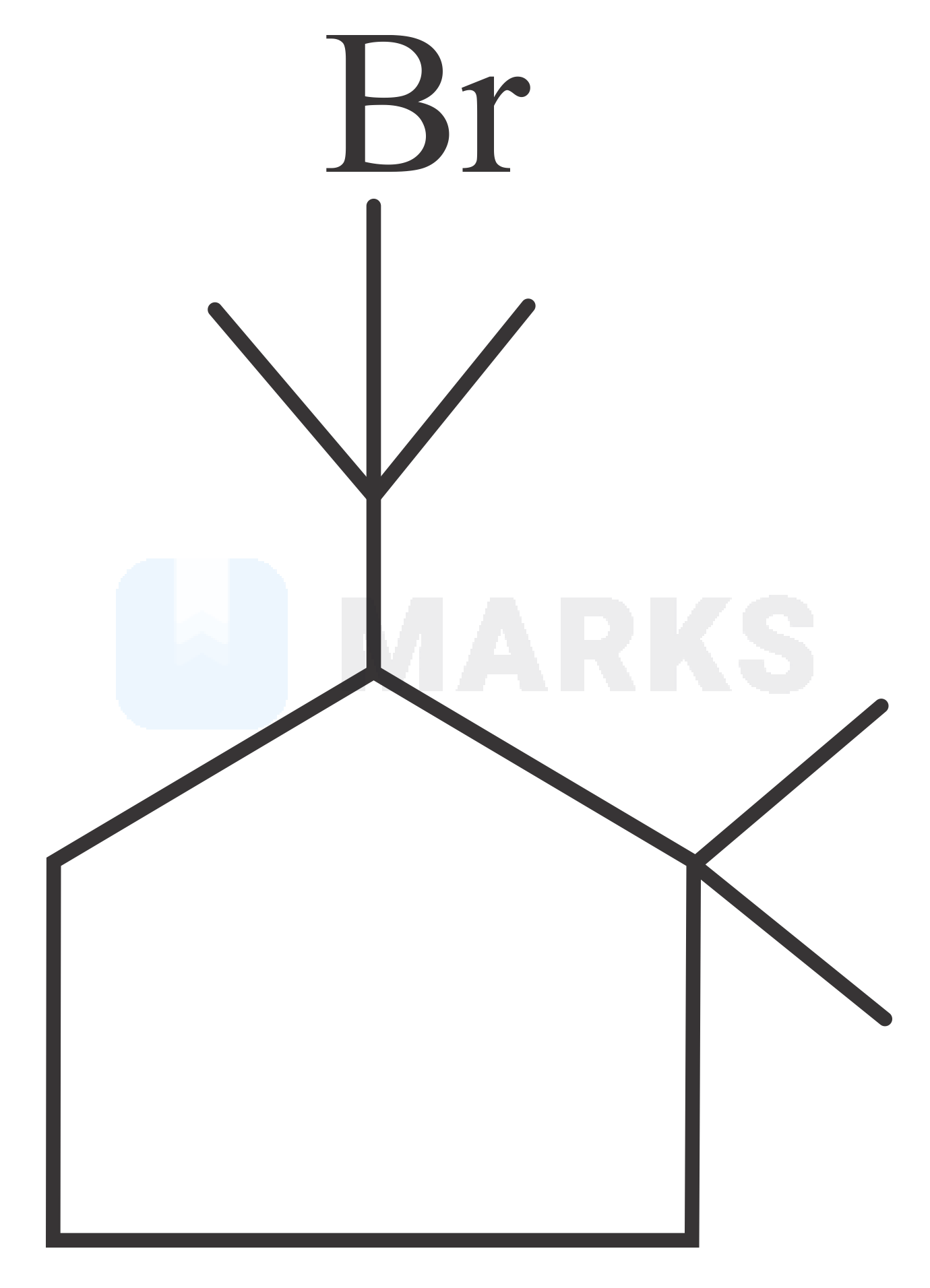
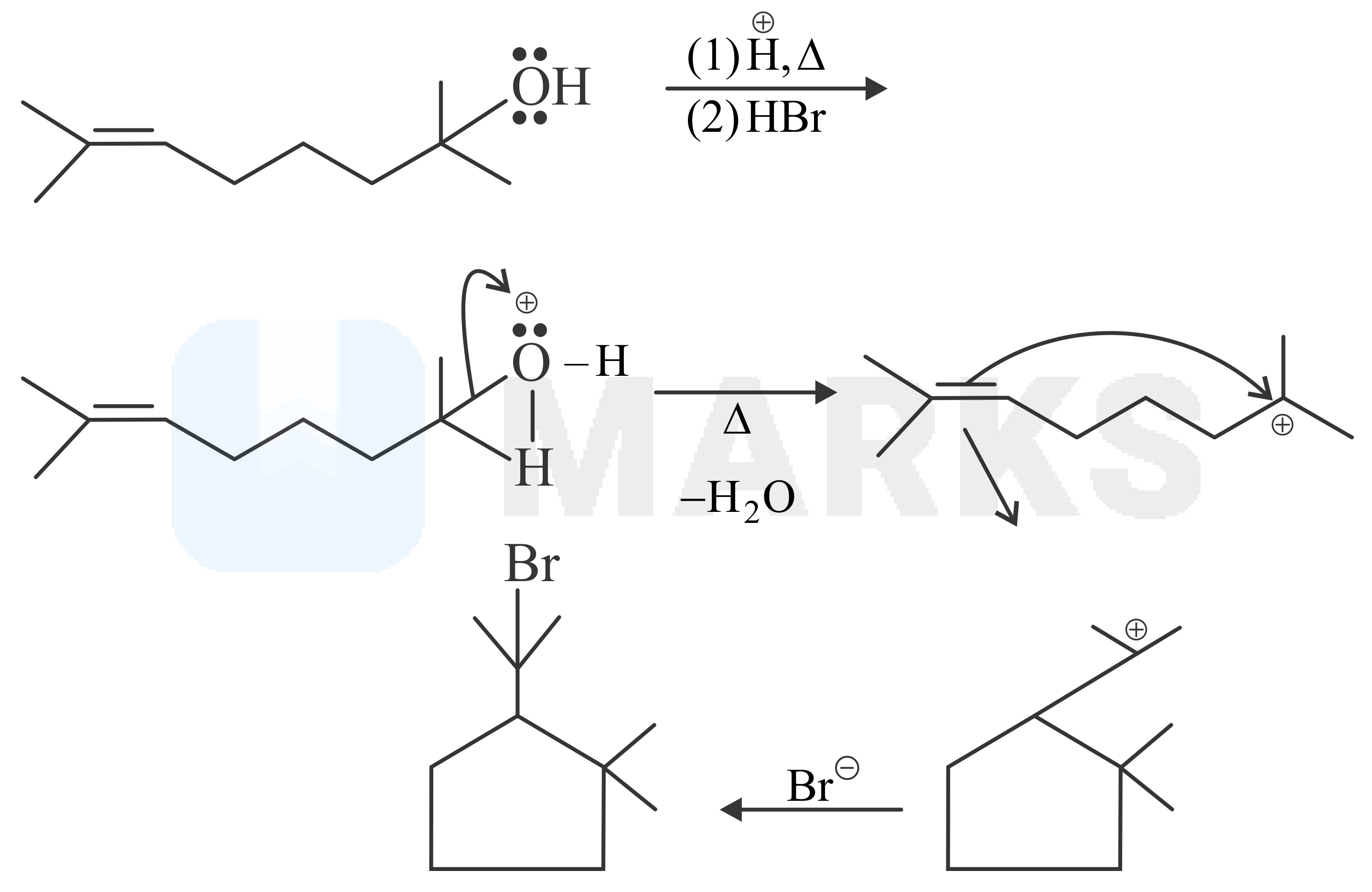
In first step of the reaction will attack on the lone pairs of electrons of the oxygen atom. Which will get dehydrated forming carbocation intermediate. On this carbocation pi bond will attack forming a five membered cyclic ring. In last step will combine with carbocation forming final product according to the following reactions.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.