Search any question & find its solution
Question:
Answered & Verified by Expert
The method of zone refining of metals is based on the principle of:
Options:
Solution:
1702 Upvotes
Verified Answer
The correct answer is:
greater solubility of the impurity in the molten state than in the solid
The principle of zone refining is that the impurities in ore of metal are more soluble in the melted state when compared to the corresponding solid state of the impurities. In the zone refining process, the impurities are concentrated at one end of the block of metal so that the rest of the block is purified.
Related Theory
Zone refining process is very effective in the removal of impurities from semiconducting elements such as Germanium, Gallium, and Silicon. This process is also used in refining high-purity metals.
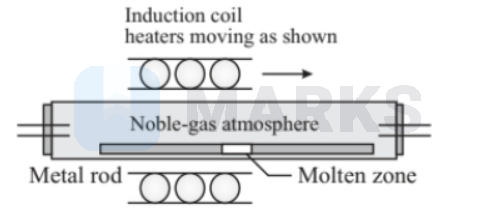
Related Theory
Zone refining process is very effective in the removal of impurities from semiconducting elements such as Germanium, Gallium, and Silicon. This process is also used in refining high-purity metals.

Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.