Search any question & find its solution
Question:
Answered & Verified by Expert
The molar solubility of $\mathrm{CaF}_2$ $\left(\mathrm{K}_{\mathrm{sp}}=5.3 \times 10^{-11}\right)$ in $0.1 \mathrm{M}$ solution of $\mathrm{NaF}$ will be
Options:
Solution:
1914 Upvotes
Verified Answer
The correct answer is:
$5.3 \times 10^{-9} \mathrm{~mol} \mathrm{~L}^{-1}$
Let the solubility of $\mathrm{CaF}_2$ in $0.1 \mathrm{M} \mathrm{NaF}$ is ' $\mathrm{S}$ ' $\mathrm{mol} \mathrm{L}^{-1}$
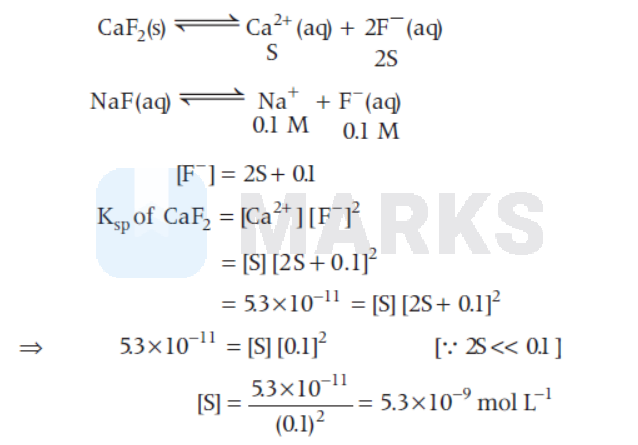
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.