Search any question & find its solution
Question:
Answered & Verified by Expert
The number of bent-shaped molecule/s from the following is ______
Solution:
1568 Upvotes
Verified Answer
The correct answer is:
3
It has hybridised central atom. Hence it is linear in shape.
(triiodide):
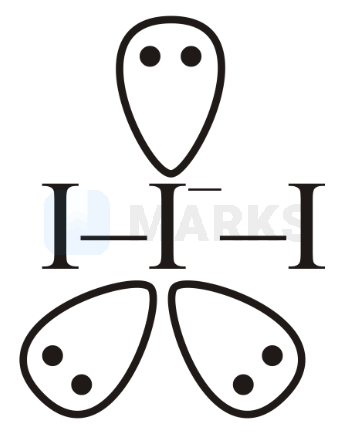
It has linear geometry, hybridisation with three lone pairs at central atom.
(nitrite) :
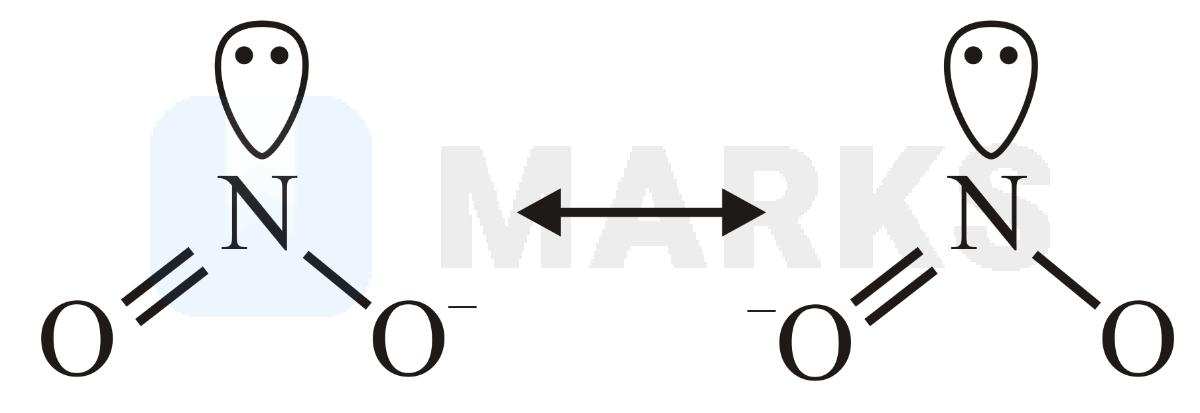
It is (nonlinear) bent shaped as it has hybridisation with one lone pair at central atom.
(ozone) :
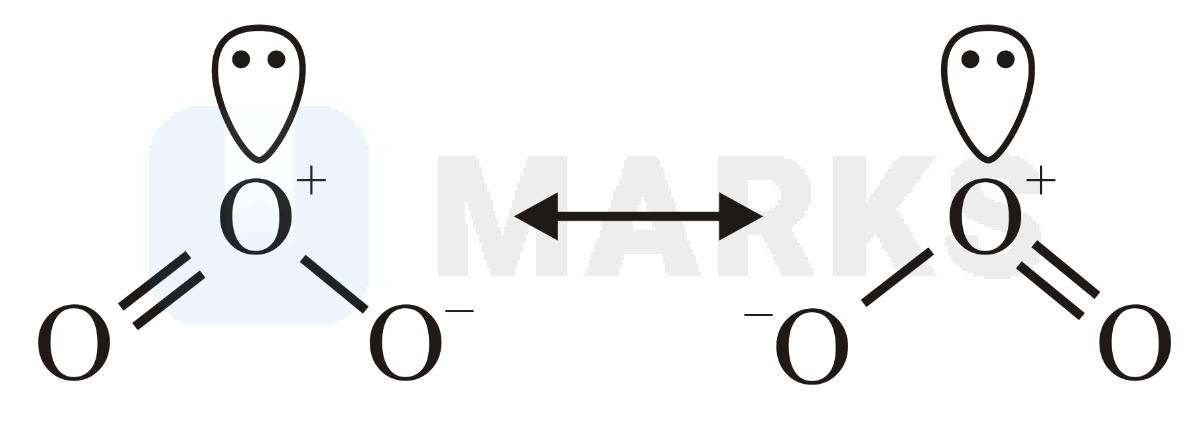
It Is bent in shape with hybridisation and one lone pair at central atom.
(sulphur dioxide) :
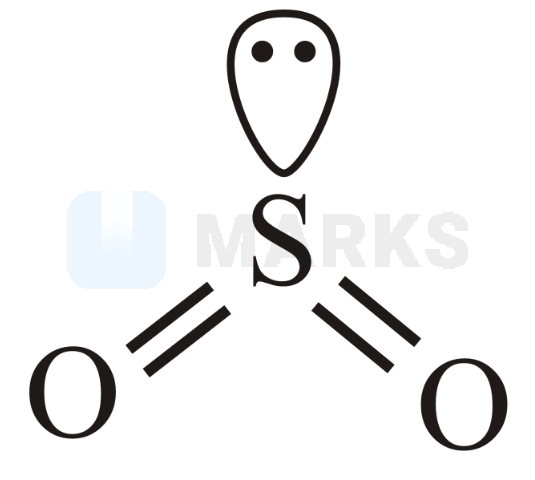
It is bent in shape with hybridisation and one lone pair at central atom.
So among the given molecules is bent in shape.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.