Search any question & find its solution
Question:
Answered & Verified by Expert
The order of acidic strength of boron trihalides
Options:
Solution:
2652 Upvotes
Verified Answer
The correct answer is:
The strength of acidic character of boron trihalides depends upon back bonding.
In boron trihalides, -back bonding occurs due to empty orbital of boron and filled orbitals of halogens.
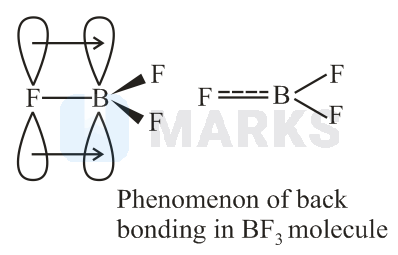
The back bonding is shown maximum by , as the size of and are small and comparatively same. Due to this effect tendency of accepting lone-pair of electrons of boron decreases as size of halogen increases. The order of size of halogens are . Thus, acidic nature is in order
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.