Search any question & find its solution
Question:
Answered & Verified by Expert
The order of basicity among the following nitrogen compounds is
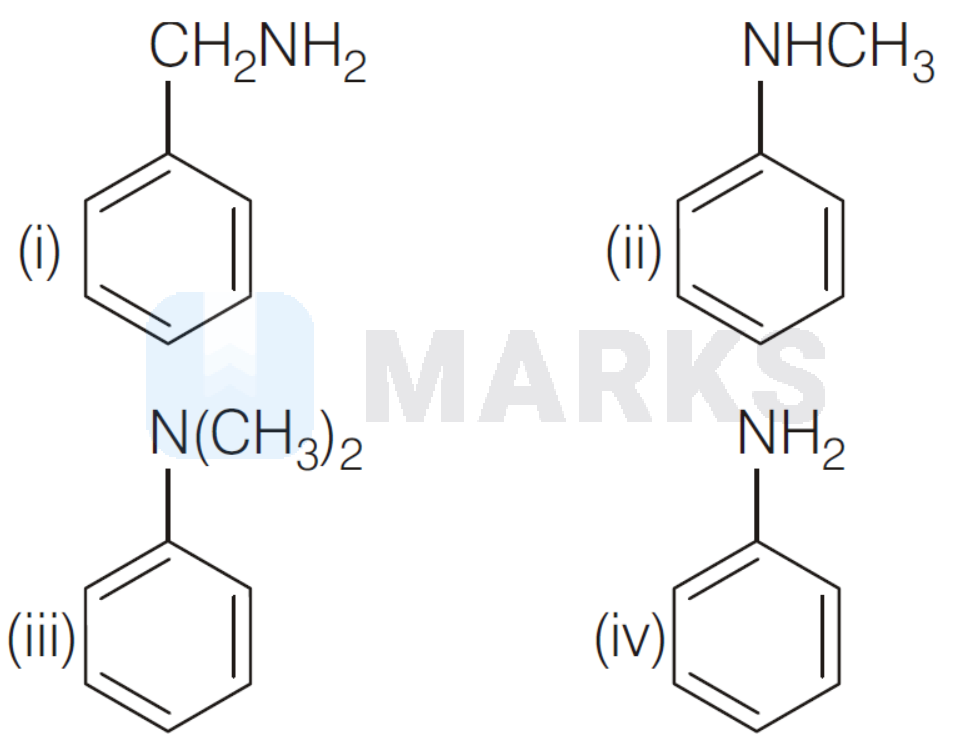
Options:

Solution:
1490 Upvotes
Verified Answer
The correct answer is:
i $>$ iii $>$ ii $>$ iv
Aryl amines have amine group directly attached to aromatic ring. These are weaker bases than arylalkyl amines due to involvement of lone pair of nitrogen in resonance with benzene ring which decreases the availability of lone pair of $e^{-}$ on nitrogen.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.