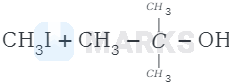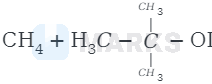Search any question & find its solution
Question:
Answered & Verified by Expert
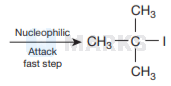
The product formed during the following reaction are
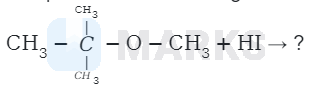
Options:
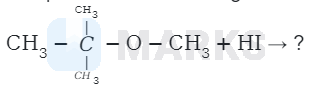
Solution:
1433 Upvotes
Verified Answer
The correct answer is:
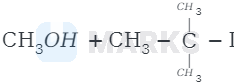
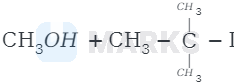
The reaction involved is
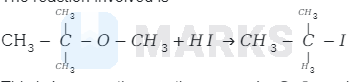
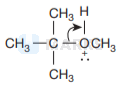
This is because the reaction occurs by \( S_{N} I \) mechanism. The formation of products is governed by the stability of the
carbocation formed from the cleavage of \( \mathrm{C}-\mathrm{O} \) bond in the protonated ether (oxonium ion). Since tert-butyl carbocation
\( \left[\left(\mathrm{CH}_{3}\right)_{3} \mathrm{C}^{+}\right] \)is more stable than the methyl carbocation \( \mathrm{CH}_{3}^{+} \)
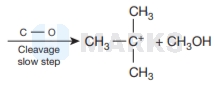
Therefore, cleavage of \( \mathrm{C}-\mathrm{O} \) bond gives a more stable carbocation \( \left[\left(\mathrm{CH}_{3}\right)_{3} \mathrm{C}^{+}\right] \)and methanol as products.
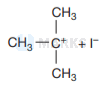
Then iodide ion \( \left(I^{-}\right) \)attacks tert-butyl carbocation to formtert-butyl iodide.
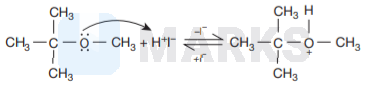
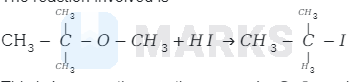
This is because the reaction occurs by \( S_{N} I \) mechanism. The formation of products is governed by the stability of the
carbocation formed from the cleavage of \( \mathrm{C}-\mathrm{O} \) bond in the protonated ether (oxonium ion). Since tert-butyl carbocation
\( \left[\left(\mathrm{CH}_{3}\right)_{3} \mathrm{C}^{+}\right] \)is more stable than the methyl carbocation \( \mathrm{CH}_{3}^{+} \)
Therefore, cleavage of \( \mathrm{C}-\mathrm{O} \) bond gives a more stable carbocation \( \left[\left(\mathrm{CH}_{3}\right)_{3} \mathrm{C}^{+}\right] \)and methanol as products.
Then iodide ion \( \left(I^{-}\right) \)attacks tert-butyl carbocation to formtert-butyl iodide.
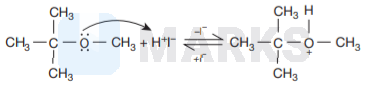




Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.