Search any question & find its solution
Question:
Answered & Verified by Expert
The products of the following reactions are
(i) \( \mathrm{C}_{6} \mathrm{H}_{5} \mathrm{~N}_{2} \mathrm{Cl}+\mathrm{Cu}_{2} \mathrm{Cl}_{2} \rightarrow \)
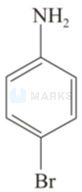
(ii) \( \mathrm{C}_{6} \mathrm{H}_{5} \mathrm{NH}_{2}+\mathrm{Br}_{2} \) (aq) \( \rightarrow \)
Options:
(i) \( \mathrm{C}_{6} \mathrm{H}_{5} \mathrm{~N}_{2} \mathrm{Cl}+\mathrm{Cu}_{2} \mathrm{Cl}_{2} \rightarrow \)
(ii) \( \mathrm{C}_{6} \mathrm{H}_{5} \mathrm{NH}_{2}+\mathrm{Br}_{2} \) (aq) \( \rightarrow \)
Solution:
2942 Upvotes
Verified Answer
The correct answer is:
\( { }_{6} \mathrm{H}_{5} \mathrm{Cl} \) and

The Cl, Br and CN nucleophiles can easily be introduced in the benzene ring of benzene diazonium salt in the presence of Cu(I) ion. This reaction is called Sandmeyer reaction.
(i)
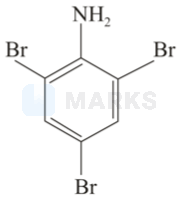
When aniline treated with bromine water, the bromine water gets decolourized and white precipitate is formed. This reaction results in the formation of 2,4,6-tribromo phenylamine.
(ii) 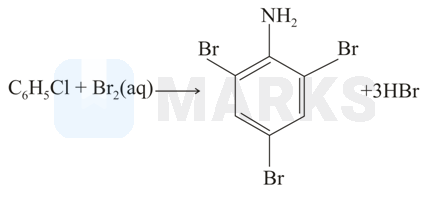
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.