Search any question & find its solution
Question:
Answered & Verified by Expert
$\mathrm{Ti}^{2+}$ is purple while $\mathrm{Ti}^{4+}$ is colourless because
Options:
Solution:
2206 Upvotes
Verified Answer
The correct answer is:
$\mathrm{Ti}^{2+}$ has $3 d^2$ configuration
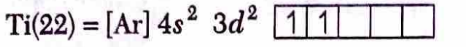
$\begin{aligned}
\mathrm{Ti}^{2+} & =[\mathrm{Ar}] 3 d^2 \\
\mathrm{Ti}^{4+} & =[\mathrm{Ar}] 3 d^0
\end{aligned}$
$\mathrm{Ti}^{2+}$ has two unpaired electrons in $3 d$ orbital and hence $d-d$ transition is possible due to absorption of light in visible region whereas $\mathrm{Ti}^{4+}$ is diamagnetic hence colourless.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.