Search any question & find its solution
Question:
Answered & Verified by Expert
Transitions between three energy levels in a particular atom give rise to three spectral lines of wavelengths, in increasing magnitudes and Which one of the following equations correctly relates and ?
Options:
Solution:
2556 Upvotes
Verified Answer
The correct answer is:
Let the three energy levels be , and . The wavelength and of the spectra lines corresponding to the three energy transitions are depicted as shown in figure.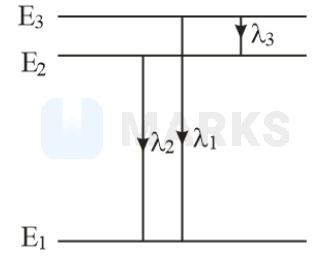
or
(given )
Thus, for the three wavelengths, we have
Now,
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.