Search any question & find its solution
Question:
Answered & Verified by Expert
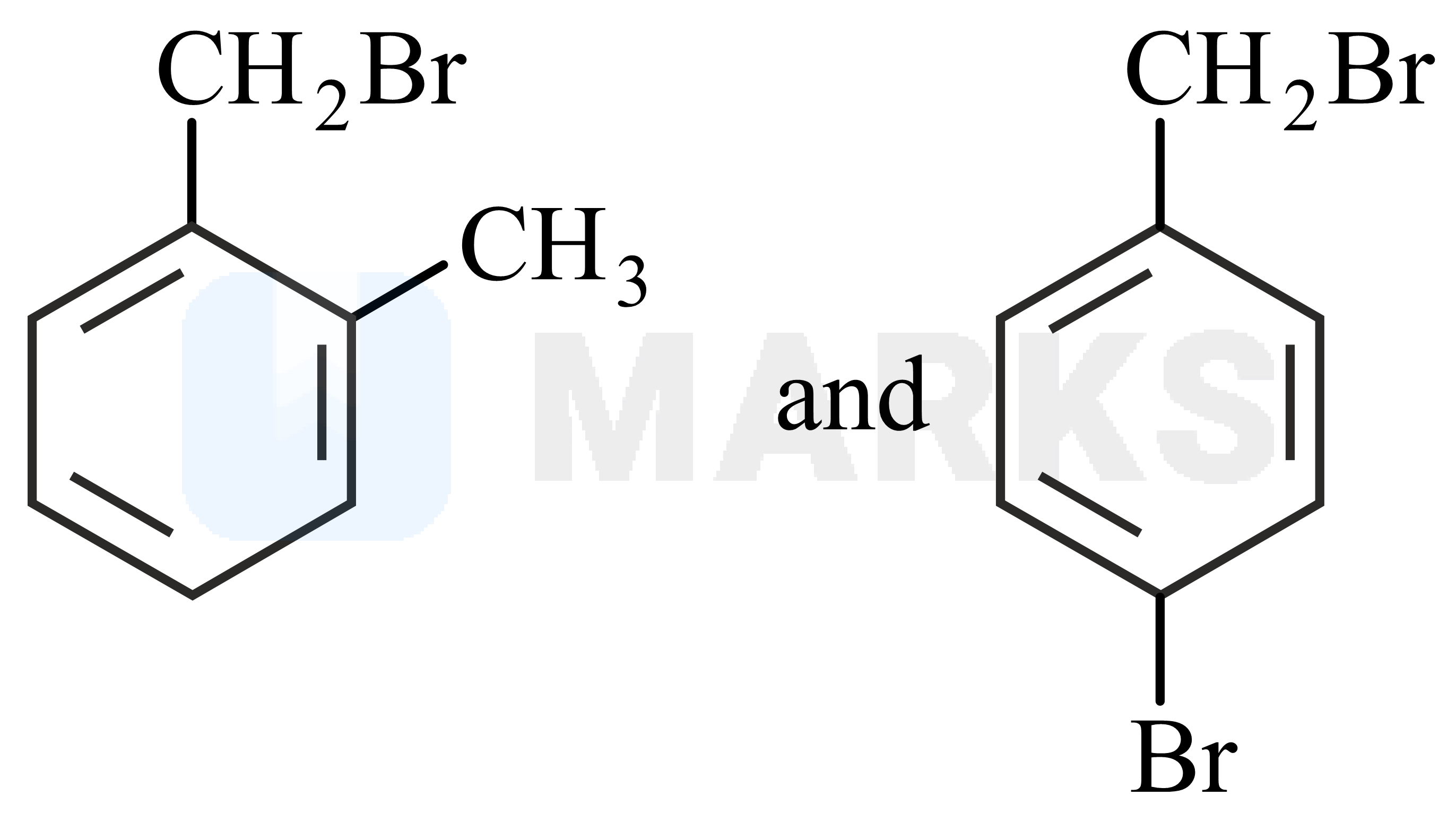
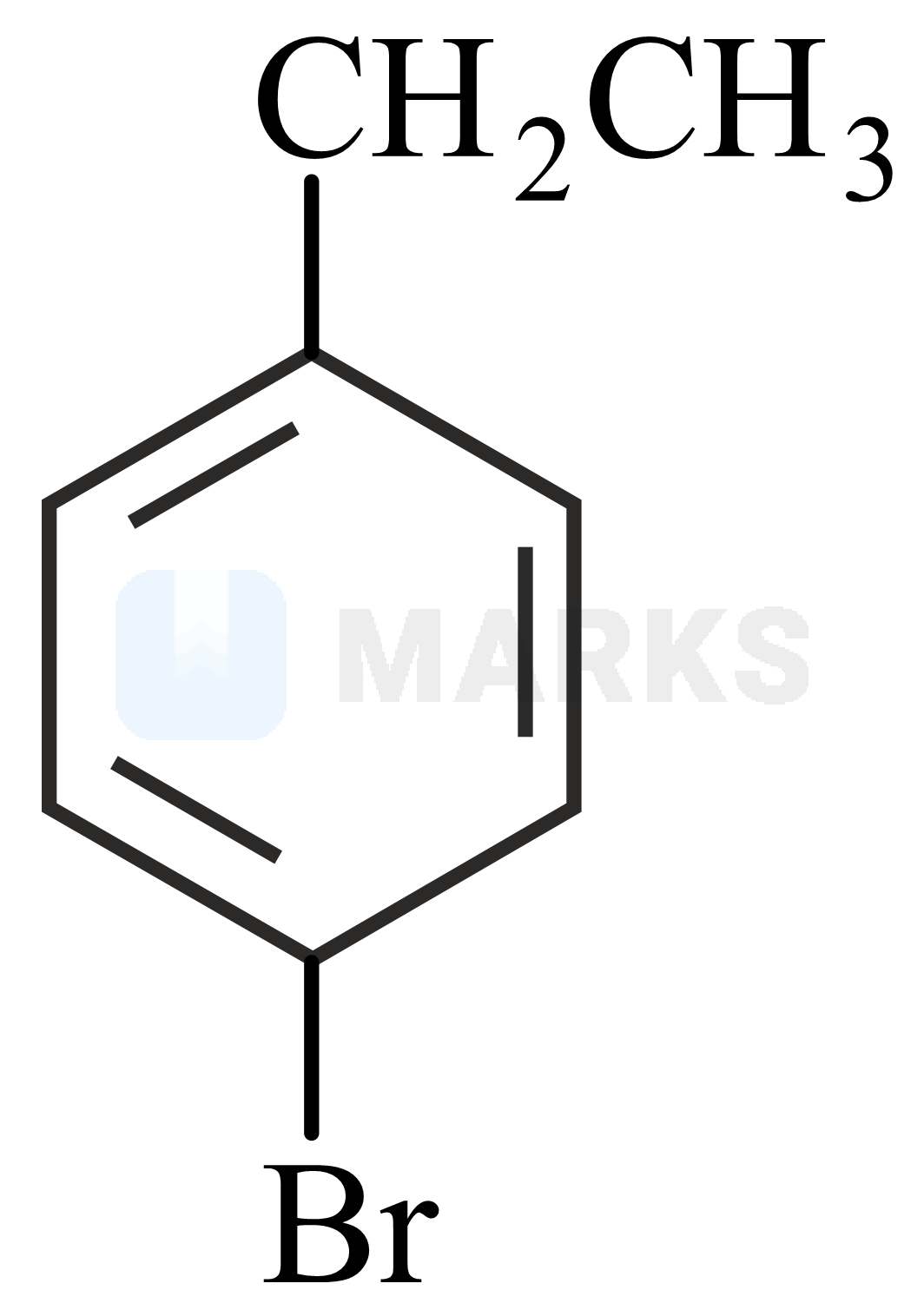
Two isomers and with Molar mass and elemental compositio and gave benzoic acid and bromobenzoic acid, respectively on oxidation with . Isomer is optically active and gives a pale yellow precipitate when warmed with alcoholic . Isomer and are, respectively :
Options:
Solution:
1948 Upvotes
Verified Answer
The correct answer is:
and 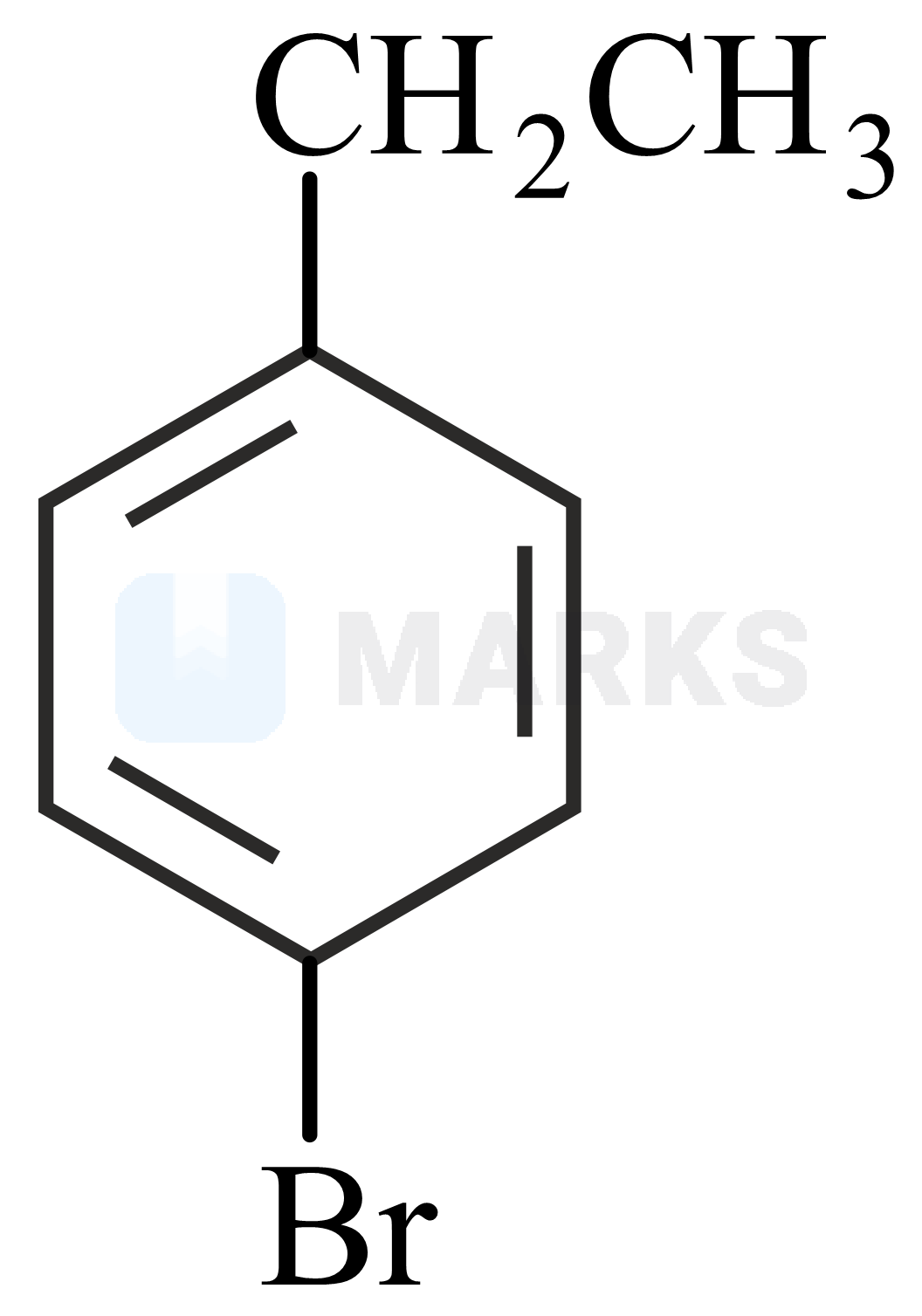
Based on the percentage composition of the elements given the molecular formula of the compound is
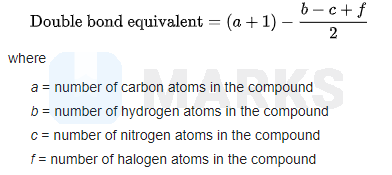
And the degree of unsaturation or double bond equivalent of is 4.
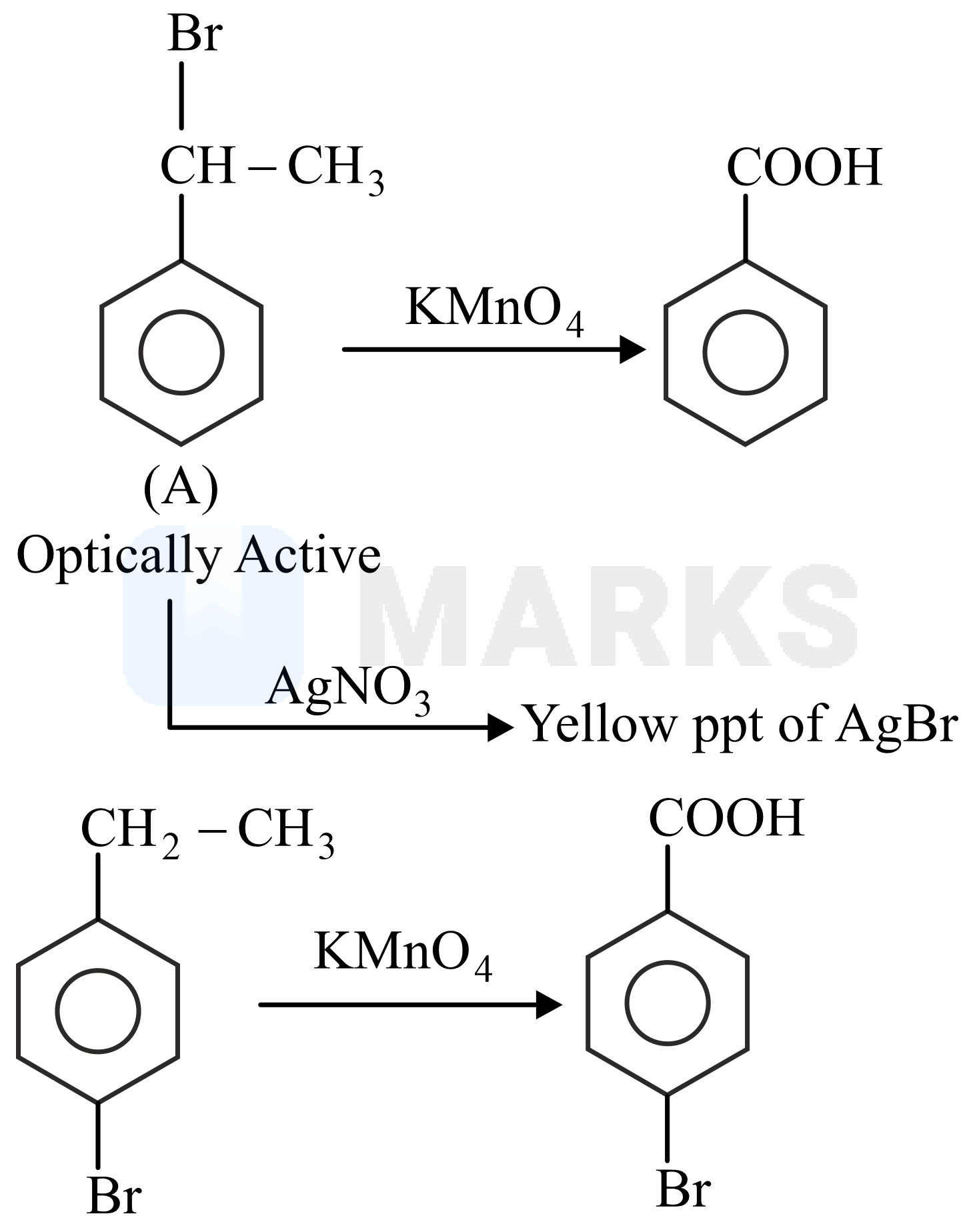
It is mentioned that A is optically active and gives pale yellow precipitate with .
Hence, the structures of A and B can be deduced based on the following reactions.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.
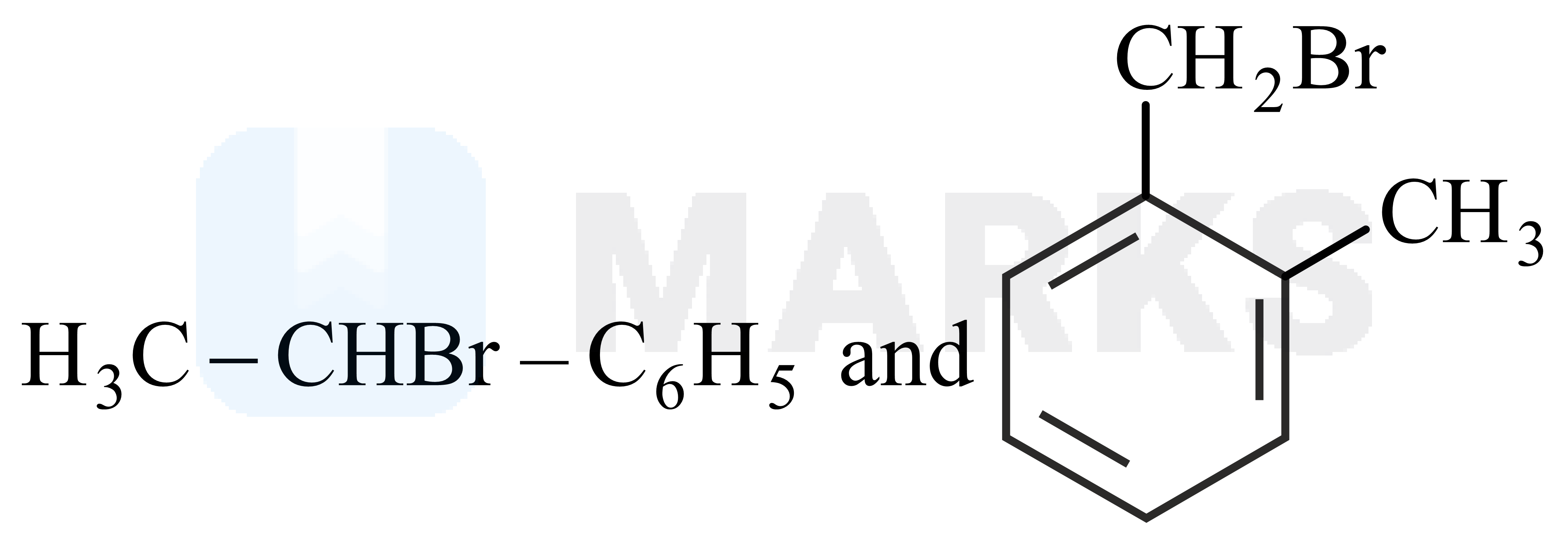
 and
and