Search any question & find its solution
Question:
Answered & Verified by Expert
Which of the following species is paramagnetic in nature
Options:
Solution:
2295 Upvotes
Verified Answer
The correct answer is:
Carbanion
Without intermediate reaction take place as under
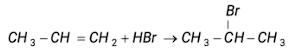
(According to markownikoff rule)
But the halogen bonded with terminal carbon so it take place in presence of peroxide by free radical mechanism.
$\underset{\text { peroxide }}{-O-O R} \rightarrow 2 R \dot{O} ; \mathrm{HBr}+\dot{\mathrm{RO}} \rightarrow \mathrm{ROH}+\mathrm{Br}$.
$\mathrm{CH}_3-\mathrm{CH}=\mathrm{CH}_2+\mathrm{Br} \rightarrow \mathrm{CH}_3-\mathrm{C} \dot{\mathrm{H}}-\mathrm{CH}_2-\mathrm{Br}$
$\mathrm{CH}_3-\mathrm{C} \mathrm{H}-\mathrm{CH}_2 \mathrm{Br}+\mathrm{HBr} \rightarrow$ $\mathrm{CH}_3-\mathrm{CH}_2-\mathrm{CH}_2 \mathrm{Br}+\mathrm{Br}$.
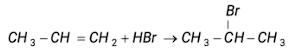
(According to markownikoff rule)
But the halogen bonded with terminal carbon so it take place in presence of peroxide by free radical mechanism.
$\underset{\text { peroxide }}{-O-O R} \rightarrow 2 R \dot{O} ; \mathrm{HBr}+\dot{\mathrm{RO}} \rightarrow \mathrm{ROH}+\mathrm{Br}$.
$\mathrm{CH}_3-\mathrm{CH}=\mathrm{CH}_2+\mathrm{Br} \rightarrow \mathrm{CH}_3-\mathrm{C} \dot{\mathrm{H}}-\mathrm{CH}_2-\mathrm{Br}$
$\mathrm{CH}_3-\mathrm{C} \mathrm{H}-\mathrm{CH}_2 \mathrm{Br}+\mathrm{HBr} \rightarrow$ $\mathrm{CH}_3-\mathrm{CH}_2-\mathrm{CH}_2 \mathrm{Br}+\mathrm{Br}$.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.