Search any question & find its solution
Question:
Answered & Verified by Expert
A Cylinder containing an ideal gas (0.1 mol of ) is in thermal equilibrium with a large volume of 0.5 molal aqueous solution of ethylene glycol at its freezing point. If the stoppers and (as shown in the figure) are suddenly withdrawn, the volume of the gas in litres after equilibrium is achieved will be
(Given, (water) = 2.0 K kg , R = 0.08 atm )
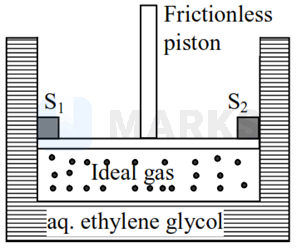
Options:
(Given, (water) = 2.0 K kg , R = 0.08 atm )

Solution:
2658 Upvotes
Verified Answer
The correct answer is:
2.18
Apply Boyle's law
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.