Search any question & find its solution
Question:
Answered & Verified by Expert
A cylindrical tube of uniform cross-sectional area $A$ is fitted with two air tight frictionless pistons. The pistons are connected to each other by a metallic wire. Initially, the pressure of the gas is $p_0$ and temperature is $T_0$, atmospheric pressure is also $p_0$. Now, the temperature of the gas is increased to $2 T_0$, the tension of wire will be
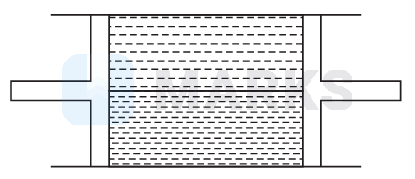
Options:

Solution:
1408 Upvotes
Verified Answer
The correct answer is:
$p_0 A$
Volume of the gas is constant i.e. $V$ = constant
$\therefore p \propto T$ i.e. pressure will be doubled if temperature is doubled.
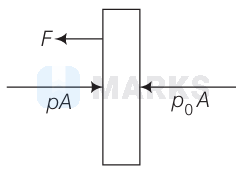
Let $F$ be the tension in the wire. Then, equilibrium of any one pipes gives.
$F=\left(p-p_0\right) A=\left(2 p_0-p_0\right) A=p_0 A$
$\therefore p \propto T$ i.e. pressure will be doubled if temperature is doubled.

Let $F$ be the tension in the wire. Then, equilibrium of any one pipes gives.
$F=\left(p-p_0\right) A=\left(2 p_0-p_0\right) A=p_0 A$
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.