Search any question & find its solution
Question:
Answered & Verified by Expert
A gas is present in a cylinder fitted with movable piston. Above and below the piston there is equal number of moles of gas. The volume above is two times the volume below at a temperature of . At what temperature will the volume above be four times the volume below-
Options:
Solution:
1714 Upvotes
Verified Answer
The correct answer is:
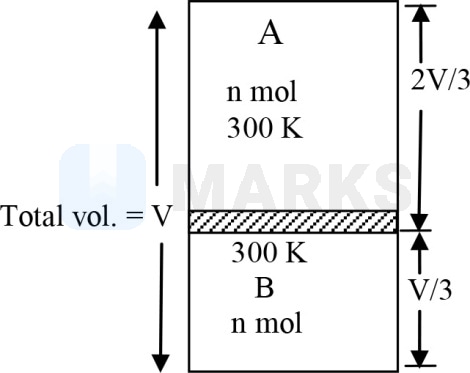
Let pressure given by
or
or
Let the final
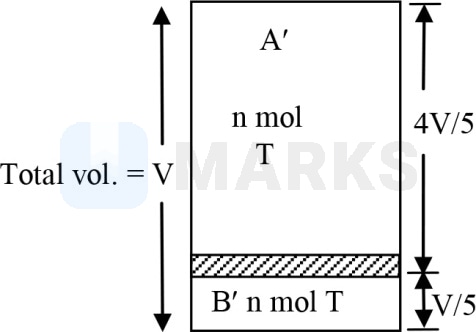
or
or
or
or
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.