Search any question & find its solution
Question:
Answered & Verified by Expert
A mixture of formic acid and oxalic acid is treated with concentrate . The evolved gaseous mixture is passed through pellets. What will be the weight in grams of the remaining product at ?
Options:
Solution:
2783 Upvotes
Verified Answer
The correct answer is:
For gases :
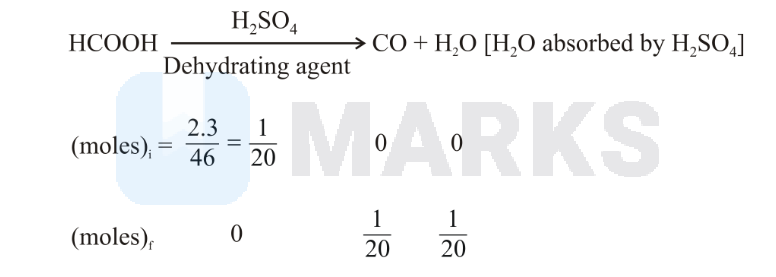
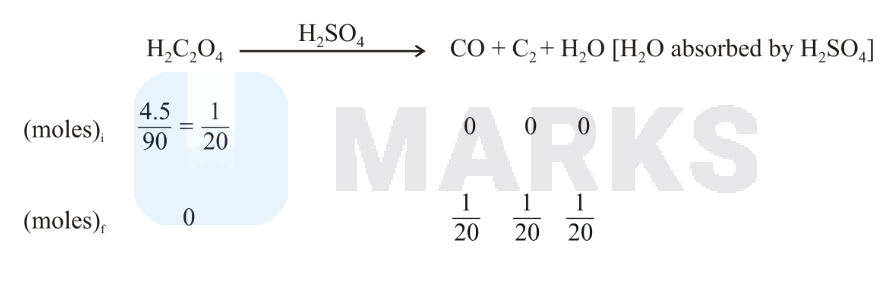
is absorbed by . So the remaning product is only . moles of formed from both reactions
Left mass of = molesmolar mass

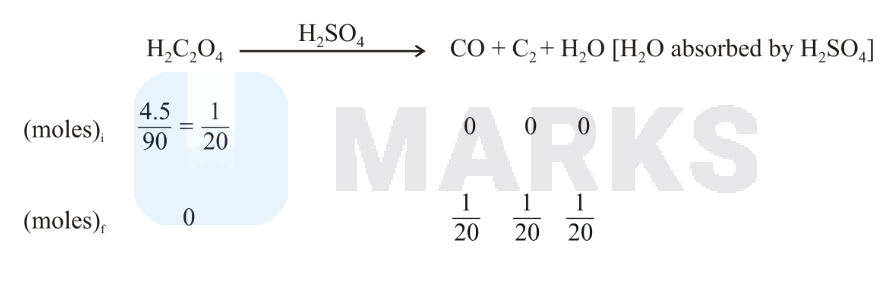
is absorbed by . So the remaning product is only . moles of formed from both reactions
Left mass of = molesmolar mass
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.