Search any question & find its solution
Question:
Answered & Verified by Expert
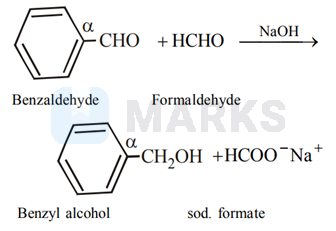
A mixture of benzaldehyde and formaldehyde on heating with aqueous $\mathrm{NaOH}$ solution gives
Options:
Solution:
2031 Upvotes
Verified Answer
The correct answer is:
benzyl alcohol and sodium formate
Benzaldyde and formaldehyde, both do not have $\alpha$ - hydrogen atom, so both will undergo Cannizzaro reaction; here formaldehyde will always be oxidised to formate while the other aldehyde $\left(\mathrm{C}_{6} \mathrm{H}_{5} \mathrm{CHO}\right.$ or any other aldehyde not having $\alpha-\mathrm{H}$, viz- $\left.\mathrm{Me}_{3} \mathrm{CCHO}\right)$ will always be reduced to corresponding alcohol (crossed Cannizzaro reaction)

Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.