Search any question & find its solution
Question:
Answered & Verified by Expert
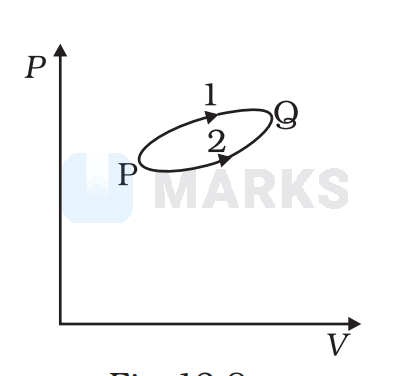
A system goes from P to Q by two different paths in the P-V diagram as shown in Fig. 12.8. Heat given to the system in path 1 is 1000 J. The work done by the system along path 1 is more than path 2 by 100 J. What is the heat exchanged by the system in path 2?
Options:

Solution:
2175 Upvotes
Verified Answer
The correct answer is:
900 J
Correct Option is : (D)
900 J
900 J
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.