Search any question & find its solution
Question:
Answered & Verified by Expert
A thermodynamic process is carried out from an original state $D$ to an intermediate state $E$ by the linear process shown in figure.
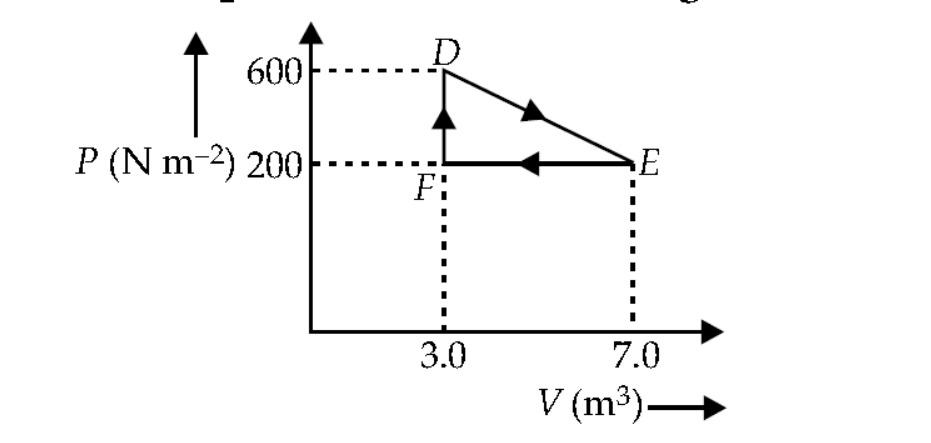
The total work is done by the gas from $D$ to $E$ to $F$ is
Options:

The total work is done by the gas from $D$ to $E$ to $F$ is
Solution:
2043 Upvotes
Verified Answer
The correct answer is:
$800 \mathrm{~J}$
Total work done by the gas from $D$ to $E$ to $F$ is equal to the area of $\triangle D E F$.
$\therefore \quad$ The area of $\triangle D E F=\frac{1}{2} D F \times E F$
Here, $D F=$ change in pressure $=600-200$
$$
=400 \mathrm{~N} \mathrm{~m}^{-2}
$$
Also, $E F=$ change in volume $=7 \mathrm{~m}^3-3 \mathrm{~m}^3$ $=4 \mathrm{~m}^3$
Area of $\triangle D E F=\frac{1}{2} \times 400 \times 4=800 \mathrm{~J}$
Thus, the total work done by the gas from $D$ to $E$ to $F$ is $800 \mathrm{~J}$.
$\therefore \quad$ The area of $\triangle D E F=\frac{1}{2} D F \times E F$
Here, $D F=$ change in pressure $=600-200$
$$
=400 \mathrm{~N} \mathrm{~m}^{-2}
$$
Also, $E F=$ change in volume $=7 \mathrm{~m}^3-3 \mathrm{~m}^3$ $=4 \mathrm{~m}^3$
Area of $\triangle D E F=\frac{1}{2} \times 400 \times 4=800 \mathrm{~J}$
Thus, the total work done by the gas from $D$ to $E$ to $F$ is $800 \mathrm{~J}$.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.