Search any question & find its solution
Question:
Answered & Verified by Expert
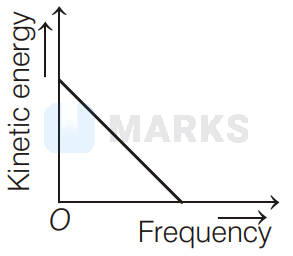
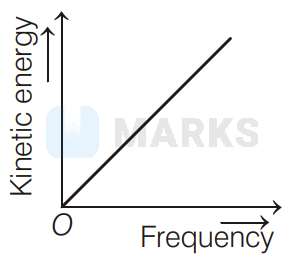
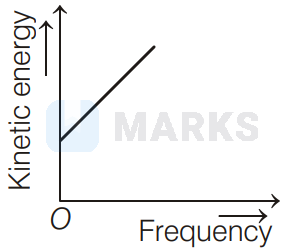
According to Einstein's photoelectric equation to the graph between kinetic energy of photoelectrons ejected and the frequency of incident radiation is
Options:
Solution:
2151 Upvotes
Verified Answer
The correct answer is:
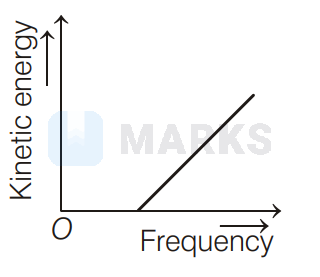

The maximum kinetic energy of photoelectron ejected is given by $\mathrm{KE}=h \mathrm{v}-\phi_{0}=h \mathrm{v}-h \mathrm{v}_{0}=h\left(\mathrm{v}-\mathrm{v}_{0}\right)$
If the frequency of incident radiation is greater than threshold frequency $v_{0}$, then the ejection of photoelectrons starts. After that as frequency increases, kinetic energy also increases linearly. It is shown in option (d).
If the frequency of incident radiation is greater than threshold frequency $v_{0}$, then the ejection of photoelectrons starts. After that as frequency increases, kinetic energy also increases linearly. It is shown in option (d).
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.