Search any question & find its solution
Question:
Answered & Verified by Expert
Among the following the molecule possessing highest dipole moment is
Options:
Solution:
1979 Upvotes
Verified Answer
The correct answer is:
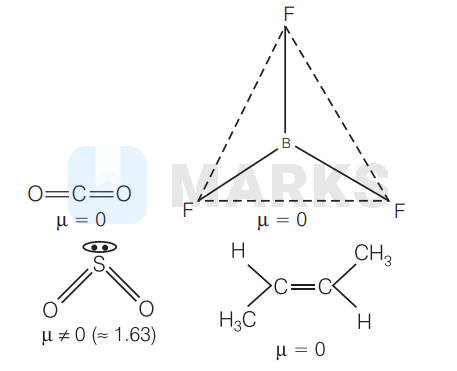
$\mathrm{SO}_2$
Among the given compounds/molecules, only $\mathrm{SO}_2$ has unsymmetrical (angular) structure, thus has the highest dipole moment.

Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.