Search any question & find its solution
Question:
Answered & Verified by Expert
Among the following the pair in which the two species are not isostructural is:
Options:
Solution:
2306 Upvotes
Verified Answer
The correct answer is:
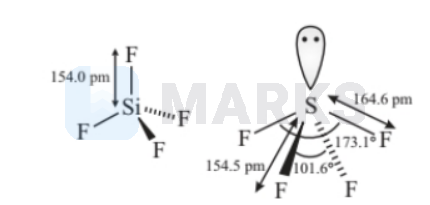
$\mathrm{SiF}_4$ and $\mathrm{SF}_4$
$\mathrm{SiF}_4$ and $\mathrm{SF}_4$ are not isostructural because $\mathrm{SiF}_4$ is tetrahedral due to $s p^3$ hybridisation while $\mathrm{SF}_4$ has distorted tetrahedral geometry due to presence of lone pair.
Related Theory
Two molecules are isostructural if they have a similar number of atoms arranged in a similar structure (spatial coordinates).
Caution
If they have same number of lone pair and bond pair, they are considered as isostructural.



Related Theory
Two molecules are isostructural if they have a similar number of atoms arranged in a similar structure (spatial coordinates).
Caution
If they have same number of lone pair and bond pair, they are considered as isostructural.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.