Search any question & find its solution
Question:
Answered & Verified by Expert
An air column closed in a tube sealed at one end by a column having height . When the tube is placed with open end down, the height of the air column is . If the tube is turned so that its open end is at the top, the height of the air column is What is the atmospheric pressure -
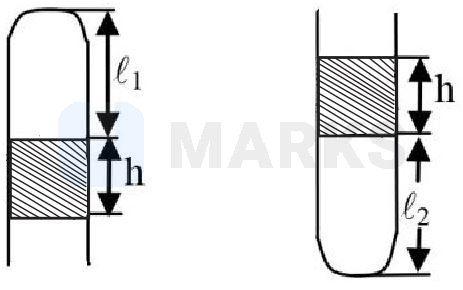
Options:

Solution:
2710 Upvotes
Verified Answer
The correct answer is:
.
Let be the value at atmospheric pressure
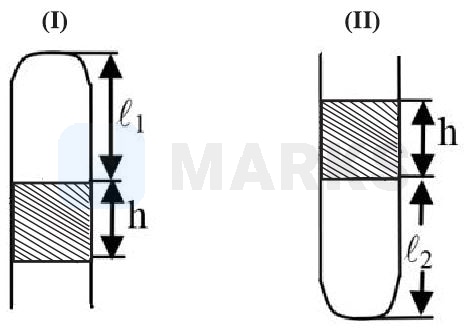
In case I:
In case ii
By applying Boyle's law between &
or
or
or

In case I:
In case ii
By applying Boyle's law between &
or
or
or
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.