Search any question & find its solution
Question:
Answered & Verified by Expert
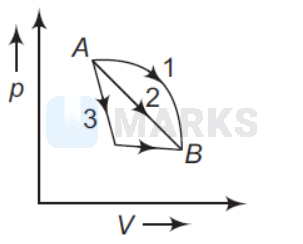
An ideal gas goes from state $A$ to state $B$ via three different processes as indicated in the $p-V$ diagram
If $Q_1, Q_2, Q_3$ indicate the heat absorbed by the gas along the three processes and $\Delta U_1, \Delta U_2, \Delta U_3$ indicate the change in internal energy along the three processes respectively, then
Options:
If $Q_1, Q_2, Q_3$ indicate the heat absorbed by the gas along the three processes and $\Delta U_1, \Delta U_2, \Delta U_3$ indicate the change in internal energy along the three processes respectively, then

Solution:
1576 Upvotes
Verified Answer
The correct answer is:
$Q_1>Q_2>Q_3$ and $\Delta U_1=\Delta U_2=\Delta U_3$
For all process 1,2 and 3
$$
\begin{array}{ll}
& \Delta U=U_B-U_A \text { is same } \\
\therefore & \Delta U_1=\Delta U_2=\Delta U_3 \\
\text { Now, } & \Delta Q=\Delta U+\Delta W
\end{array}
$$
Now, $\Delta W=$ work done by the gas
$$
\therefore \quad \Delta Q_1>\Delta Q_2>\Delta Q_3
$$
$$
\begin{array}{ll}
& \Delta U=U_B-U_A \text { is same } \\
\therefore & \Delta U_1=\Delta U_2=\Delta U_3 \\
\text { Now, } & \Delta Q=\Delta U+\Delta W
\end{array}
$$
Now, $\Delta W=$ work done by the gas
$$
\therefore \quad \Delta Q_1>\Delta Q_2>\Delta Q_3
$$
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.