Search any question & find its solution
Question:
Answered & Verified by Expert
An ideal gas is expanded from to under different conditions. The incorrect statement among the following is?
Options:
Solution:
2244 Upvotes
Verified Answer
The correct answer is:
The change in internal energy of the gas is (i) zero, if it is expanded reversibly with and (ii) positive, if it is expanded reversibly under adiabatic conditions with
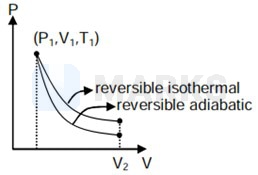
Area under curve in reversible isothermal is more. So, more work will be done by gas.
In reversible adiabatic expansion,
So ,
In free expansion,
So
If carried out isothermally
(adiabatic); from I law
If carried out adiabatically
(isothermal); From I law
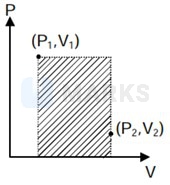
During irreversible compression, maximum work is done on the gas (corresponding to shaded area).
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.