Search any question & find its solution
Question:
Answered & Verified by Expert
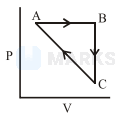
An ideal gas undergoes a cyclic process as shown in Figure.
$\begin{aligned}
&\Delta \mathrm{U}_{\mathrm{BC}}=-5 \mathrm{~kJ} \mathrm{~mol}^{-1}, \mathrm{q}_{\mathrm{AB}}=2 \mathrm{~kJ} \mathrm{~mol}^{-1} \\
&\mathrm{~W}_{\mathrm{AB}}=-5 \mathrm{~kJ} \mathrm{~mol}^{-1}, \mathrm{~W}_{\mathrm{CA}}=3 \mathrm{~kJ} \mathrm{~mol}^{-1}
\end{aligned}$
Heat absorbed by the system during process CA is:
Options:

$\begin{aligned}
&\Delta \mathrm{U}_{\mathrm{BC}}=-5 \mathrm{~kJ} \mathrm{~mol}^{-1}, \mathrm{q}_{\mathrm{AB}}=2 \mathrm{~kJ} \mathrm{~mol}^{-1} \\
&\mathrm{~W}_{\mathrm{AB}}=-5 \mathrm{~kJ} \mathrm{~mol}^{-1}, \mathrm{~W}_{\mathrm{CA}}=3 \mathrm{~kJ} \mathrm{~mol}^{-1}
\end{aligned}$
Heat absorbed by the system during process CA is:
Solution:
2957 Upvotes
Verified Answer
The correct answer is:
$+5 \mathrm{~kJ} \mathrm{~mol}^{-1}$
$+5 \mathrm{~kJ} \mathrm{~mol}^{-1}$
$\Delta \mathrm{U}_{\mathrm{AB}}=\mathrm{q}_{\mathrm{AB}}+\mathrm{W}_{\mathrm{AB}}=2+(-5)=-3 \mathrm{~kJ} / \mathrm{mol}$
$\Delta \mathrm{U}_{\mathrm{BC}}=-5 \mathrm{~kJ} / \mathrm{mol}$
For cyclic process, $\Delta \mathrm{U}=0$
$\begin{aligned}
&\Delta \mathrm{U}_{\mathrm{AB}}+\Delta \mathrm{U}_{\mathrm{BC}}+\Delta \mathrm{U}_{\mathrm{CA}}=0 \\
&\Delta \mathrm{U}_{\mathrm{CA}}=-\Delta \mathrm{U}_{\mathrm{AB}}-\Delta \mathrm{U}_{\mathrm{BC}} \\
&\Delta \mathrm{U}_{\mathrm{CA}}=-(-3)-(-5)=8 \mathrm{~kJ} / \mathrm{mol} \\
&\Delta \mathrm{U}_{\mathrm{CA}}=\mathrm{q}_{\mathrm{CA}}+\mathrm{W}_{\mathrm{CA}} \\
&8=\mathrm{q}_{\mathrm{CA}}+3 \\
&\mathrm{q}_{\mathrm{CA}}=+5 \mathrm{~kJ} / \mathrm{mol}
\end{aligned}$
Heat absorbed has positive sign.
$\Delta \mathrm{U}_{\mathrm{BC}}=-5 \mathrm{~kJ} / \mathrm{mol}$
For cyclic process, $\Delta \mathrm{U}=0$
$\begin{aligned}
&\Delta \mathrm{U}_{\mathrm{AB}}+\Delta \mathrm{U}_{\mathrm{BC}}+\Delta \mathrm{U}_{\mathrm{CA}}=0 \\
&\Delta \mathrm{U}_{\mathrm{CA}}=-\Delta \mathrm{U}_{\mathrm{AB}}-\Delta \mathrm{U}_{\mathrm{BC}} \\
&\Delta \mathrm{U}_{\mathrm{CA}}=-(-3)-(-5)=8 \mathrm{~kJ} / \mathrm{mol} \\
&\Delta \mathrm{U}_{\mathrm{CA}}=\mathrm{q}_{\mathrm{CA}}+\mathrm{W}_{\mathrm{CA}} \\
&8=\mathrm{q}_{\mathrm{CA}}+3 \\
&\mathrm{q}_{\mathrm{CA}}=+5 \mathrm{~kJ} / \mathrm{mol}
\end{aligned}$
Heat absorbed has positive sign.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.