Search any question & find its solution
Question:
Answered & Verified by Expert
An unsaturated hydrocarbon 'A' adds two molecules of $\mathrm{H}_2$ and on reductive ozonolysis gives butane-1, 4-dial, ethanal and propanone. Give the structure of ' $A$ ', write its IUPAC name and explain the reactions involved.
Solution:
1789 Upvotes
Verified Answer
Two molecules of hydrogen add on 'A' this shows that ' $\mathrm{A}$ ' is either an alkadiene or an alkyne.
On reductive ozonolysis ' $A$ ' gives three fragments, one of which is dialdehyde. Hence, the molecule has broken down at two sites. Therefore, 'A' has two double bonds. It gives the following three fragments:
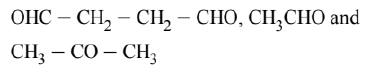
Hence, its structure as deduced from the three fragments must be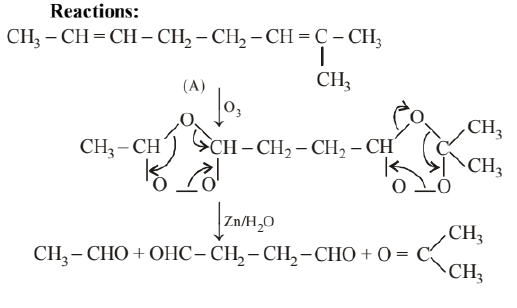
On reductive ozonolysis ' $A$ ' gives three fragments, one of which is dialdehyde. Hence, the molecule has broken down at two sites. Therefore, 'A' has two double bonds. It gives the following three fragments:
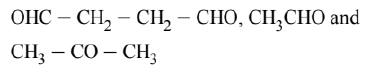
Hence, its structure as deduced from the three fragments must be

Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.