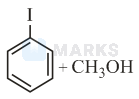Search any question & find its solution
Question:
Answered & Verified by Expert
Anisole on cleavage with gives
Options:
Solution:
2427 Upvotes
Verified Answer
The correct answer is:
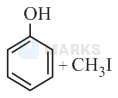

Anisole is methoxy benzene. Anisole reacts with protons from hydroiodic acid to form methyl(phenyl) oxonium ion.
In the methyl(phenyl)oxonium ion, the oxygen atom has a positive charge. This positive charge is delocalised through resonance with the benzene ring. Due to this, the bond between the oxygen atom and the aromatic carbon atom of benzene has a partial double bond character. This bond is difficult to break. Hence, the iodide ion does not attack the aromatic carbon atom of the benzene ring. On the other hand, the bond between the oxygen atom and the carbon atom of the methyl group is a single bond. This bond can be easily broken.
Hence, the iodide ion attacks the carbon atom of the methyl group to form phenol and iodomethane.
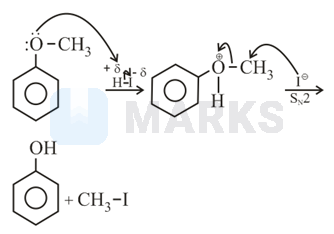
In the methyl(phenyl)oxonium ion, the oxygen atom has a positive charge. This positive charge is delocalised through resonance with the benzene ring. Due to this, the bond between the oxygen atom and the aromatic carbon atom of benzene has a partial double bond character. This bond is difficult to break. Hence, the iodide ion does not attack the aromatic carbon atom of the benzene ring. On the other hand, the bond between the oxygen atom and the carbon atom of the methyl group is a single bond. This bond can be easily broken.
Hence, the iodide ion attacks the carbon atom of the methyl group to form phenol and iodomethane.

Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.