Search any question & find its solution
Question:
Answered & Verified by Expert
Arrange the following in decreasing order of their acidic strength. Give explanation for the arrangement.
$\mathrm{C}_6 \mathrm{H}_5 \mathrm{COOH}, \mathrm{FCH}_2 \mathrm{COOH}, \mathrm{NO}_2 \mathrm{CH}_2 \mathrm{COOH}$
$\mathrm{C}_6 \mathrm{H}_5 \mathrm{COOH}, \mathrm{FCH}_2 \mathrm{COOH}, \mathrm{NO}_2 \mathrm{CH}_2 \mathrm{COOH}$
Solution:
1808 Upvotes
Verified Answer
.$$
\text { Electron withdrawing ability of } \mathrm{NO}_2, \mathrm{~F} \text { and } \mathrm{C}_6 \mathrm{H}_5 \text { are as follows: }
$$
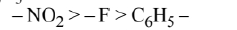
$$
\text { Therefore, decreasing order of their acidic strength is given as }
$$
$$
\mathrm{NO}_2 \mathrm{CH}_2 \mathrm{COOH}>\mathrm{FCH}_2 \mathrm{COOH}>\mathrm{C}_6 \mathrm{H}_5 \mathrm{COOH}
$$
\text { Electron withdrawing ability of } \mathrm{NO}_2, \mathrm{~F} \text { and } \mathrm{C}_6 \mathrm{H}_5 \text { are as follows: }
$$
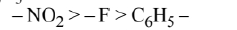
$$
\text { Therefore, decreasing order of their acidic strength is given as }
$$
$$
\mathrm{NO}_2 \mathrm{CH}_2 \mathrm{COOH}>\mathrm{FCH}_2 \mathrm{COOH}>\mathrm{C}_6 \mathrm{H}_5 \mathrm{COOH}
$$
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.