Search any question & find its solution
Question:
Answered & Verified by Expert
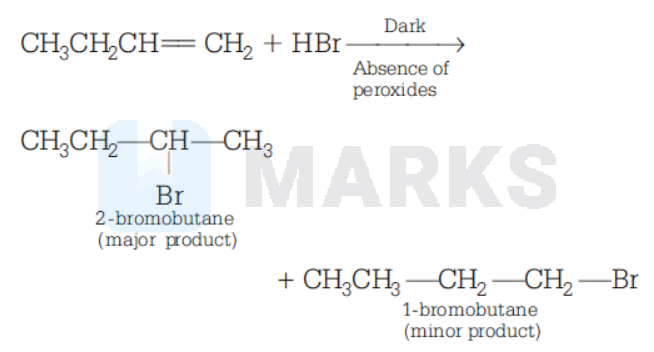
Assertion (A): Reaction of 1-butene with $\mathrm{HBr}$ gives 1-bromobutane as major product.
Reason (R) Addition of hydrogen halides to alkenes proceeds according to Markownikoff's rule.
The correct answer is
Options:
Reason (R) Addition of hydrogen halides to alkenes proceeds according to Markownikoff's rule.
The correct answer is
Solution:
2036 Upvotes
Verified Answer
The correct answer is:
$\mathrm{A}$ is not correct but $\mathrm{R}$ is correct
According to Markownikoff's rule, the addition of reagents such as $\mathrm{HX}, \mathrm{H}_2 \mathrm{O}$ and $\mathrm{HOX}$ with unsymmetrical alkanes occurs in such a way that the negative part of the adding molecule goes to that carbon atom of the double bond which carries lesser number of hydrogen atoms.

Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.