Search any question & find its solution
Question:
Answered & Verified by Expert
Assertion : $\left[\mathrm{Fe}\left(\mathrm{H}_2 \mathrm{O}\right)_5 \mathrm{NO}\right] \mathrm{SO}_4$ is paramagnetic.Reason : The Fe in $\left[\mathrm{Fe}\left(\mathrm{H}_2 \mathrm{O}\right)_5 \mathrm{NO}\right] \mathrm{NO}_4$ has three unpaired electrons.
Options:
Solution:
2159 Upvotes
Verified Answer
The correct answer is:
If both assertion and reason are true and reason is the correct explanation of assertion
(a) : $\mathrm{Fe}^{+}:[\mathrm{Ar}] 3 d^6 4 s^1$
When the weak field ligand $\mathrm{H}_2 \mathrm{O}$ and strong field ligand NO attack, the configuration changes as follows:
$\mathrm{Fe}^{+}:[\mathrm{Ar}] 3 d^7 4 s^0$
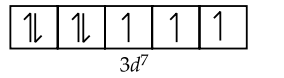
$\therefore \quad \mathrm{Fe}^{+}$has 3 unpaired electrons.
When the weak field ligand $\mathrm{H}_2 \mathrm{O}$ and strong field ligand NO attack, the configuration changes as follows:
$\mathrm{Fe}^{+}:[\mathrm{Ar}] 3 d^7 4 s^0$

$\therefore \quad \mathrm{Fe}^{+}$has 3 unpaired electrons.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.