Search any question & find its solution
Question:
Answered & Verified by Expert
Atomic number of $\mathrm{Cr}, \mathrm{Fe}$ and $\mathrm{Co}$ are 24, 26 and 27 respectively. Which of the following inner orbital octahedral complexes are paramagnetic?
Options:
Solution:
2759 Upvotes
Verified Answer
The correct answer is:
$\left[\mathrm{Cr}(\mathrm{CN})_6\right]^{3-}$
$\left[\mathrm{Cr}(\mathrm{CN})_6\right]^{3-}$
Oxidation state of $\mathrm{Cr}: x+(-1) 6=-3$
$$
\begin{aligned}
x & =+3 \\
\mathrm{Cr} & =[\mathrm{Ar}] 4 s^1 3 d^5 \\
\mathrm{Cr}^{3+} & =[\mathrm{Ar}] 4 s^0 3 d^3
\end{aligned}
$$
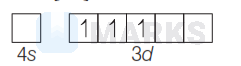
Since, $\mathrm{CN}$ is a strong field ligand, it will cause pairing of electrons in $\mathrm{Cr}^{3+}$,
So, there is one unpaired electron in $3 d$-orbital of $\mathrm{Cr}^{3+}$ which makes the compound $\left[\mathrm{Cr}(\mathrm{CN})_6\right]^{3-}$ paramagnetic.
Oxidation state of $\mathrm{Cr}: x+(-1) 6=-3$
$$
\begin{aligned}
x & =+3 \\
\mathrm{Cr} & =[\mathrm{Ar}] 4 s^1 3 d^5 \\
\mathrm{Cr}^{3+} & =[\mathrm{Ar}] 4 s^0 3 d^3
\end{aligned}
$$

Since, $\mathrm{CN}$ is a strong field ligand, it will cause pairing of electrons in $\mathrm{Cr}^{3+}$,

So, there is one unpaired electron in $3 d$-orbital of $\mathrm{Cr}^{3+}$ which makes the compound $\left[\mathrm{Cr}(\mathrm{CN})_6\right]^{3-}$ paramagnetic.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.