Search any question & find its solution
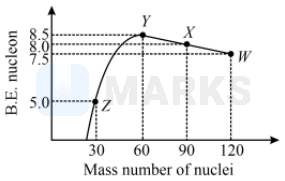
Energy is released in a process where the binding energy of the products is greater than the binding energy of the reactants.
Binding energy is given by
For,
Total binding energy of the reactants
Total binding energy of the products
Binding energy of the product is lesser than that of the reactants.
For,
Total binding energy of the reactants
Total binding energy of the products
Here, also the binding energy of the product is lesser than that of the reactants.
For,
Total binding energy of the reactants
Total binding energy of the products
Here, energy is released as the binding energy of the product is greater than that of the reactants.
For,
Total binding energy of reactants
Total binding energy of products
Here, also binding energy of the product is lesser than that of the reactants.
So, energy is released in
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.