Search any question & find its solution
Question:
Answered & Verified by Expert
Calculate the degree of ionization of \(0.05 \mathrm{M}\) acetic acid if its \(\mathrm{pK}_{\mathrm{a}}\) value is 4.74. How is the degree of dissociation affected when its solution also contains (a) \(0.01 \mathrm{M}\) (b) 0.1 \(\mathrm{M}\) in HCl?
Solution:
2813 Upvotes
Verified Answer
\(\mathrm{pK}_{\mathrm{a}}=4.74\), i. e., \(-\log \mathrm{K}_{\mathrm{a}}=4.74\)
or \(\quad \log \mathrm{K}_{\mathrm{a}}=-4.74\)
Taking Antilog \(\mathrm{K}_{\mathrm{a}}=\) Antilog \((-4.74)\)
\(\therefore \mathrm{K}_{\mathrm{a}}=1.82 \times 10^{-5}\)
\(\alpha^2=\mathrm{K}_{\mathrm{a}} / \mathrm{c}=\left(1.82 \times 10^{-5}\right) /\left(5 \times 10^{-2}\right)=1.908 \times 10^{-2}\)
In presence of \(\mathrm{HCl}\), due to high concentration of \(\mathrm{H}^{+}\)ion, dissociation equilibrium will shift backward, i.e. dissociation of acetic acid will decrease,
(a) In presence of \(0.01 \mathrm{M} \mathrm{HCl}\), if \(\mathrm{x}\) is the amount dissociated, then
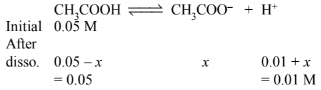
\(\left(0.01 \mathrm{M} \mathrm{H}^{+}\right.\)ions are obtained from \(\left.0.01 \mathrm{M} \mathrm{HCl}\right)\)
\((\because x< < 0.01 \quad \therefore 0.01+x \approx 0.01)\)
\(\begin{aligned}
&\therefore \mathrm{K}_{\mathrm{a}}=\frac{\left[\mathrm{CH}_3 \mathrm{COO}^{-}\right]\left[\mathrm{H}^{+}\right]}{\left[\mathrm{CH}_3 \mathrm{COOH}^{-}\right.} \\
&\therefore \alpha=\left(1.82 \times 10^{-5}\right) / 10^{-2}=1.82 \times 10^{-3} \\
&\quad\left(\text { since } \alpha=\frac{\text { Amount dissociated }}{\text { amount taken }}\right)
\end{aligned}\)
(b) In the presence of \(0.1 \mathrm{M} \mathrm{HCl}\), if \(\mathrm{y}\) is the amount of acetic acid dissociated, then at equilibrium
\(\left[\mathrm{CH}_3 \mathrm{COOH}\right]=0.05-\mathrm{y} \approx 0.05 \mathrm{M}\)
\(\begin{aligned}
&{\left[\mathrm{CH}_3 \mathrm{COO}^{-}\right]=y,\left[\mathrm{H}^{+}\right]=0.1 \mathrm{M}+\mathrm{y}=0.1 \mathrm{M}} \\
&\mathrm{K}_{\mathrm{a}}=(0.1) / 0.05 \text { or } \mathrm{y} / 0.05=\mathrm{K}_{\mathrm{a}} / 0.1 \\
&\therefore \alpha=\left(1.82 \times 10^{-5}\right) / 10^{-1}=1.82 \times 10^{-4},
\end{aligned}\)
i. e. \(\alpha=1.82 \times 10^{-4}\)
or \(\quad \log \mathrm{K}_{\mathrm{a}}=-4.74\)
Taking Antilog \(\mathrm{K}_{\mathrm{a}}=\) Antilog \((-4.74)\)
\(\therefore \mathrm{K}_{\mathrm{a}}=1.82 \times 10^{-5}\)
\(\alpha^2=\mathrm{K}_{\mathrm{a}} / \mathrm{c}=\left(1.82 \times 10^{-5}\right) /\left(5 \times 10^{-2}\right)=1.908 \times 10^{-2}\)
In presence of \(\mathrm{HCl}\), due to high concentration of \(\mathrm{H}^{+}\)ion, dissociation equilibrium will shift backward, i.e. dissociation of acetic acid will decrease,
(a) In presence of \(0.01 \mathrm{M} \mathrm{HCl}\), if \(\mathrm{x}\) is the amount dissociated, then
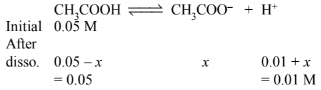
\(\left(0.01 \mathrm{M} \mathrm{H}^{+}\right.\)ions are obtained from \(\left.0.01 \mathrm{M} \mathrm{HCl}\right)\)
\((\because x< < 0.01 \quad \therefore 0.01+x \approx 0.01)\)
\(\begin{aligned}
&\therefore \mathrm{K}_{\mathrm{a}}=\frac{\left[\mathrm{CH}_3 \mathrm{COO}^{-}\right]\left[\mathrm{H}^{+}\right]}{\left[\mathrm{CH}_3 \mathrm{COOH}^{-}\right.} \\
&\therefore \alpha=\left(1.82 \times 10^{-5}\right) / 10^{-2}=1.82 \times 10^{-3} \\
&\quad\left(\text { since } \alpha=\frac{\text { Amount dissociated }}{\text { amount taken }}\right)
\end{aligned}\)
(b) In the presence of \(0.1 \mathrm{M} \mathrm{HCl}\), if \(\mathrm{y}\) is the amount of acetic acid dissociated, then at equilibrium
\(\left[\mathrm{CH}_3 \mathrm{COOH}\right]=0.05-\mathrm{y} \approx 0.05 \mathrm{M}\)
\(\begin{aligned}
&{\left[\mathrm{CH}_3 \mathrm{COO}^{-}\right]=y,\left[\mathrm{H}^{+}\right]=0.1 \mathrm{M}+\mathrm{y}=0.1 \mathrm{M}} \\
&\mathrm{K}_{\mathrm{a}}=(0.1) / 0.05 \text { or } \mathrm{y} / 0.05=\mathrm{K}_{\mathrm{a}} / 0.1 \\
&\therefore \alpha=\left(1.82 \times 10^{-5}\right) / 10^{-1}=1.82 \times 10^{-4},
\end{aligned}\)
i. e. \(\alpha=1.82 \times 10^{-4}\)
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.