Search any question & find its solution
Question:
Answered & Verified by Expert
Calculate the oxidation number of sulphur, chromium and nitrogen in \(\mathrm{H}_2 \mathrm{SO}_5, \mathrm{Cr}_2 \mathrm{O}_7^{2-}\) and \(\mathrm{NO}_3^{-}\). Suggest structure of these three compounds. Count for the fallacy.
Solution:
2949 Upvotes
Verified Answer
(i) By conventional method, the O. N. of S in \(\mathrm{H}_2 \mathrm{SO}_5\) is \(2(+1)+x+5(-2)=0\) or \(x=+8\)
This is impossible because the maximum O.N. of S cannot be more than six since it has only six electrons in the valence shell. This fallacy is overcome if we calculate the O.N. of S by chemical bonding method.
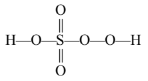
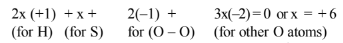
(ii) Oxidation number of chromium in \(\mathrm{Cr}_2 \mathrm{O}_7^{2-}\) :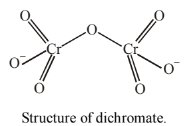
Let the Oxidation number of chromium \(=x\)
\(\begin{aligned}
&\therefore 2 x+7(-2)=-2 \Rightarrow 2 x-14=-2 \\
&\Rightarrow 2 x=-2+14 \Rightarrow 2 x=+12 \\
&\Rightarrow x=+6
\end{aligned}\)
Thus the oxidation number of chromium \(=+6\)
(iii) \(\mathrm{O}, \mathrm{N}\), of \(\mathrm{N}_{\text {in } \mathrm{NO}_3^{-}}^{-}\)
According to conventional method, O. N. of \(\mathrm{N}\) in \(\mathrm{NO}_3^{-}=\mathrm{x}+3(-2)=-1\) or \(x=+5\)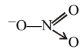
According to chemical bonding method,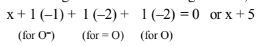
Thus, there is no fallacy about the O. N. of \(\mathrm{N}\) in \(\mathrm{NO}_3{ }^{-}\)whether one calculates by conventional method or by chemical bonding method.
This is impossible because the maximum O.N. of S cannot be more than six since it has only six electrons in the valence shell. This fallacy is overcome if we calculate the O.N. of S by chemical bonding method.

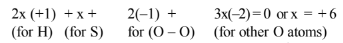
(ii) Oxidation number of chromium in \(\mathrm{Cr}_2 \mathrm{O}_7^{2-}\) :

Let the Oxidation number of chromium \(=x\)
\(\begin{aligned}
&\therefore 2 x+7(-2)=-2 \Rightarrow 2 x-14=-2 \\
&\Rightarrow 2 x=-2+14 \Rightarrow 2 x=+12 \\
&\Rightarrow x=+6
\end{aligned}\)
Thus the oxidation number of chromium \(=+6\)
(iii) \(\mathrm{O}, \mathrm{N}\), of \(\mathrm{N}_{\text {in } \mathrm{NO}_3^{-}}^{-}\)
According to conventional method, O. N. of \(\mathrm{N}\) in \(\mathrm{NO}_3^{-}=\mathrm{x}+3(-2)=-1\) or \(x=+5\)

According to chemical bonding method,
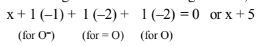
Thus, there is no fallacy about the O. N. of \(\mathrm{N}\) in \(\mathrm{NO}_3{ }^{-}\)whether one calculates by conventional method or by chemical bonding method.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.