Search any question & find its solution
A. All group elements form oxides of general formula and where and . Both the types of oxides are acidic in nature.
B. is an oxidising agent while is reducing in nature.
C. The reducing property decreases from to down the group.
D. The ozone molecule contains five lone pairs of electrons.
Choose the correct answer from the options given below:
(A) All group elements form oxides of the and type where or . And these oxides produces acids on hydrolysis.
(B) is reducing while is an oxidising agent. An oxidising agent is a substance that causes oxidation by accepting electrons; therefore, it gets reduced. A reducing agent is a substance that causes reduction by losing electrons; therefore, it gets oxidised.
(C) The reducing property increases from to down the group. This is because the bond energy bond decreases down the group.
(D) 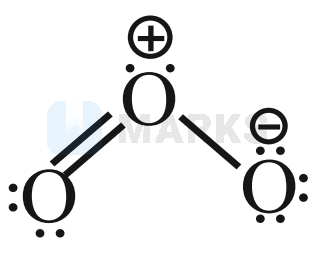 have six lone pairs
have six lone pairs
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.