Search any question & find its solution
For which molecules among the following, the resultant dipole moment ?
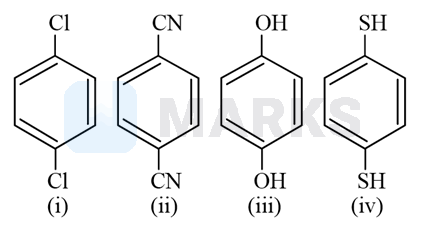
So we know the concept of dipole moment that it is depends on the charge and distance between central atom and side atoms
But the charge is more prominent then distance
It is determining as polar and non polar molecules
It is used for covalent or ionic molecule
It is for non polar molecule
it is for polar molecule
So from above theory we conclude that
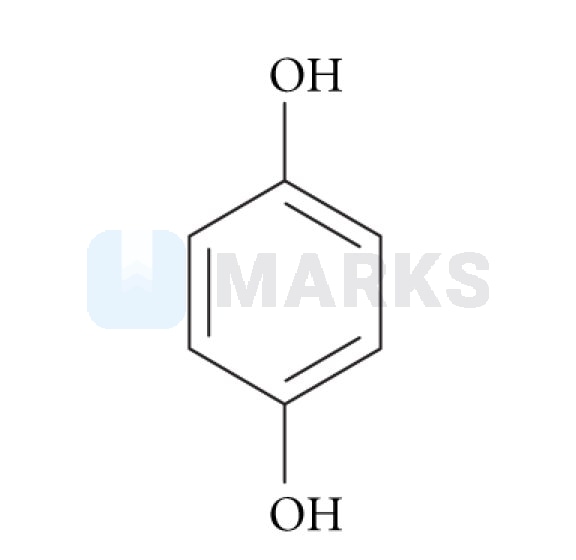
So in above hydro quinone molecule the molecule are in the position of right side that's why it's dipole moment is not be equal to zero
Also it is happend in the case of ipso-parathiophenone
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.