Search any question & find its solution
Question:
Answered & Verified by Expert
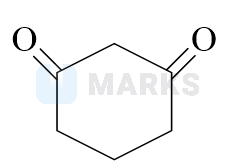
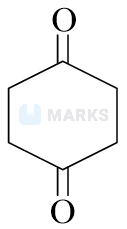
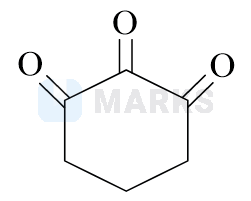
Highest enol content will be shown by :
Options:
Solution:
1129 Upvotes
Verified Answer
The correct answer is:
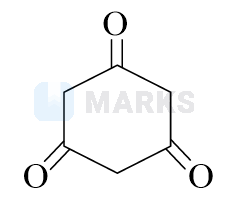

When keto form loses a proton from the alpha carbon, it goes to the enol form. Depending on whether the proton is lost from oxygen or carbon, this conjugate acid can deliver either the carbonyl form or the enol form.
In this case, after enolisation, it forms an aromatic compound.
Aromatic compounds are stable due to their aromaticity. The resonance stabilisation resulting from delocalised pi electrons distributes the charge over the entire ring, reducing the overall energy of the compound.
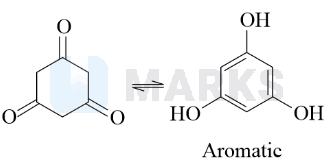
Hence, the answer is option B.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.