Search any question & find its solution
Question:
Answered & Verified by Expert
In the reaction,
$\begin{aligned} 2 \mathrm{FeSO}_{4}+\mathrm{H}_{2} \mathrm{SO}_{4}+\mathrm{H}_{2} \mathrm{O}_{2} & \longrightarrow \\ & \mathrm{Fe}_{2}\left(\mathrm{SO}_{4}\right)_{3}+2 \mathrm{H}_{2} \mathrm{O} \end{aligned}$
the oxidising agent is
Options:
$\begin{aligned} 2 \mathrm{FeSO}_{4}+\mathrm{H}_{2} \mathrm{SO}_{4}+\mathrm{H}_{2} \mathrm{O}_{2} & \longrightarrow \\ & \mathrm{Fe}_{2}\left(\mathrm{SO}_{4}\right)_{3}+2 \mathrm{H}_{2} \mathrm{O} \end{aligned}$
the oxidising agent is
Solution:
2860 Upvotes
Verified Answer
The correct answer is:
$\mathrm{H}_{2} \mathrm{O}_{2}$
Hydrogen peroxide is a powerful oxidant in acids as well as alkaline medium. It oxidises acidified ferrows sulphate to ferric sulphate.
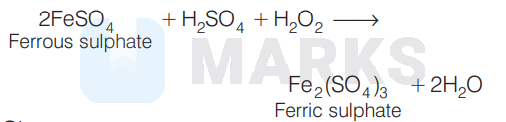
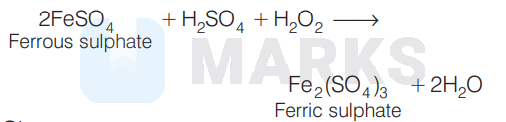
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.