Search any question & find its solution
Question:
Answered & Verified by Expert
$K_p$ for the reaction $A \rightleftharpoons B$ is 4 . If initially only $A$ is present then what will be the partial pressure of $B$ after equilibrium?
Options:
Solution:
2476 Upvotes
Verified Answer
The correct answer is:
$0.8$
Assumingthatreaction isstarted with $1 \mathrm{~mol}$ of $A$ and at equilibrium $x$ moles of $A$ have reacted,
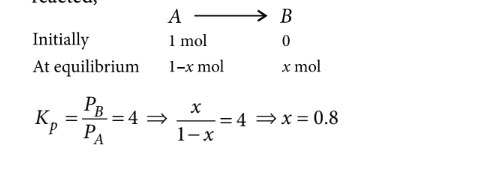
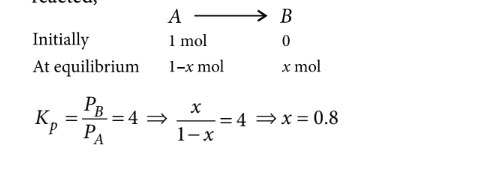
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.