Search any question & find its solution
Question:
Answered & Verified by Expert
Look at the graph
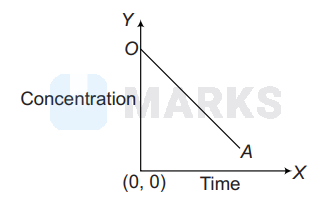
Choose the correct equation from the following which best suited to the above graph
Options:

Choose the correct equation from the following which best suited to the above graph
Solution:
2484 Upvotes
Verified Answer
The correct answer is:
$\left[A_t\right]=[A]_0+K t$
The equation is $\left[A_t\right]=\left[A_0\right]+K t$
On comparing it with general straight line equation, $Y=m x+c$
$\begin{aligned} \text { Slope }(m) & =+K \\ \text { intercept }(c) & =\left[A_0\right]\end{aligned}$
Therefore,the graph is
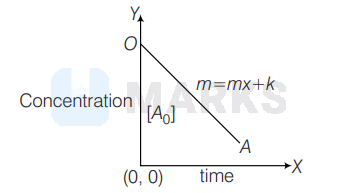
On comparing it with general straight line equation, $Y=m x+c$
$\begin{aligned} \text { Slope }(m) & =+K \\ \text { intercept }(c) & =\left[A_0\right]\end{aligned}$
Therefore,the graph is

Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.