Search any question & find its solution
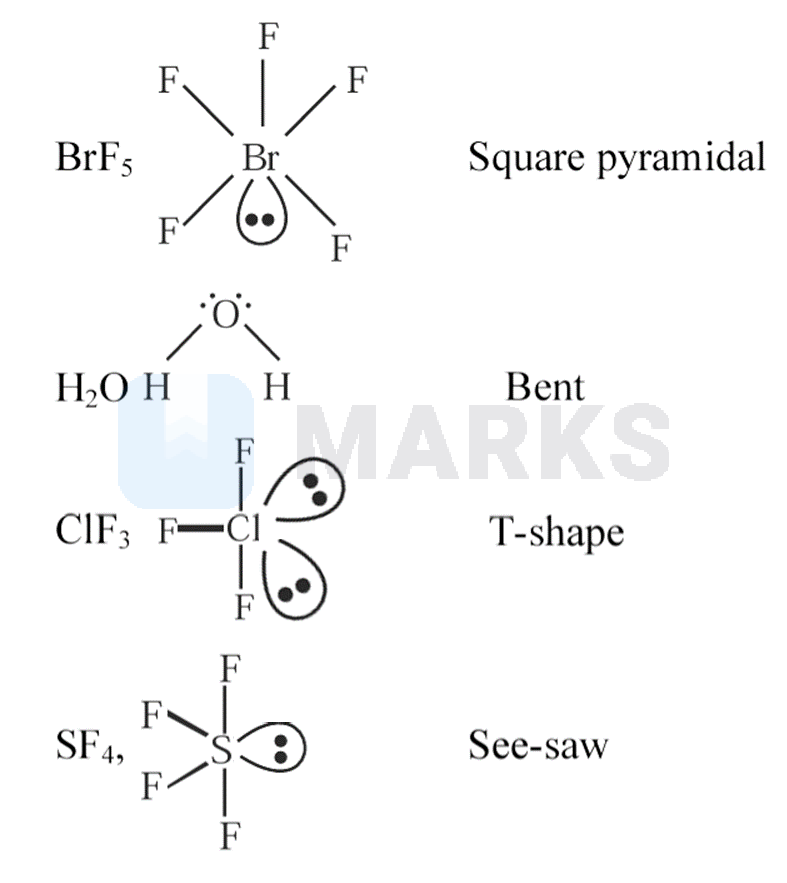
| List I (Molecule) | List II (Shape) | ||
| A. | i. | T-shape | |
| B. | ii. | See saw | |
| C. | iii. | Bent | |
| D. | iv. | Square pyramidal |
(A) The steric number of the central Bromine atom is bond pairsone lone pair . Hence the shape is square pyramidal structure and the hybridisation is .
(B) molecular geometry is bent because it has two bound atoms and two lone pairs on oxygen, the central atom. The two lone pairs on the oxygen atom push the two hydrogen atoms down further away from the oxygen atom and closer to each other, creating a bent shape and degrees angle between the two atoms.
(C) The molecular geometry of is said to be T-shaped. acquires such a shape because two lone pairs take up equatorial positions as they require more space and greater repulsions. They are arranged in a trigonal bipyramidal shape with bonds and lone pairs.
(D) has an type of hybridisation. In a molecule of , each atom is bonded to four fluorine atoms.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.