Search any question & find its solution
Question:
Answered & Verified by Expert
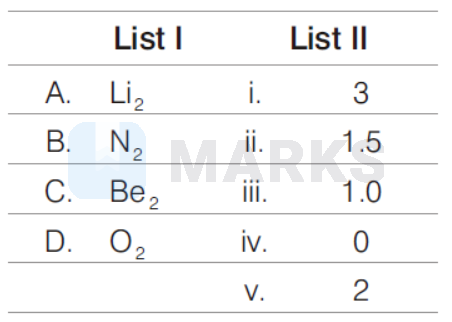
Match the bond order for the following molecules.
The correct answer is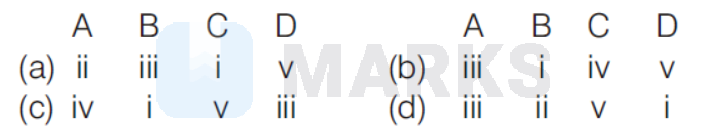
Options:

The correct answer is
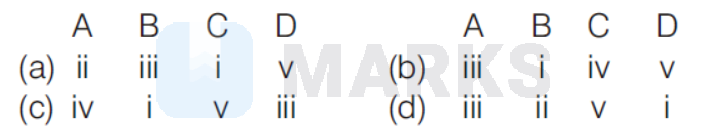
Solution:
1815 Upvotes
Verified Answer
The correct answer is:
(b)
Bond order the number of chemical bonds between a pair of atom. So,
(B.O)
$$
\begin{aligned}
& \mathrm{Li}_2=\mathrm{Li}-\mathrm{Li} \quad(1) \\
& \mathrm{N}_2=\mathrm{N} \equiv \mathrm{N}(3) \\
& \mathrm{Be}_2=\mathrm{Be} \cdot \operatorname{Be}(\text { No bond) }(0) \\
& \mathrm{O}_2=\mathrm{O}=\mathrm{O}
\end{aligned}
$$
Note Zero bond order means the molecule is too unstable and so it will not exist.
Electrons will fill according to the energy levels of the orbitals.
(B.O)
$$
\begin{aligned}
& \mathrm{Li}_2=\mathrm{Li}-\mathrm{Li} \quad(1) \\
& \mathrm{N}_2=\mathrm{N} \equiv \mathrm{N}(3) \\
& \mathrm{Be}_2=\mathrm{Be} \cdot \operatorname{Be}(\text { No bond) }(0) \\
& \mathrm{O}_2=\mathrm{O}=\mathrm{O}
\end{aligned}
$$
Note Zero bond order means the molecule is too unstable and so it will not exist.
Electrons will fill according to the energy levels of the orbitals.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.