Search any question & find its solution
Question:
Answered & Verified by Expert
Match the following

Options:

Solution:
1323 Upvotes
Verified Answer
The correct answer is:
(A) Q, (B) Q, (C) R,S, (D) P,S
(A) Q, (B) Q, (C) R,S, (D) P,S
$$
\text { (A) Match with (Q) }
$$
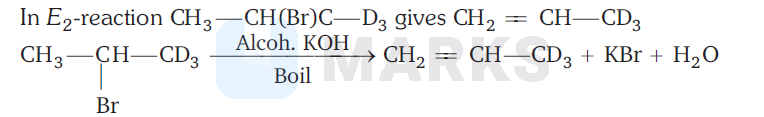
The formation of $\mathrm{CH}_2=\mathrm{CH}-\mathrm{CD}_3$ can be explained on the basis of the fact that $\mathrm{C}-\mathrm{D}$ bond is much stronger than $\mathrm{C}-\mathrm{H}$ bond.
(B) match with (Q)
Reactivity of $\mathrm{PhCHBrCH}_3$ is greater than
$\mathrm{Ph} \mathrm{CHBrCD}_3$ because $\mathrm{C}-\mathrm{D}$ bond is more stronger than $\mathrm{C}-\mathrm{H}$ bond.
$$
\text { (C) match with (R) and (S) }
$$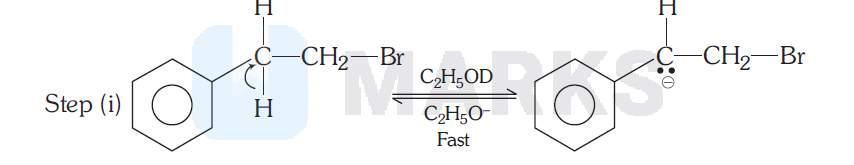
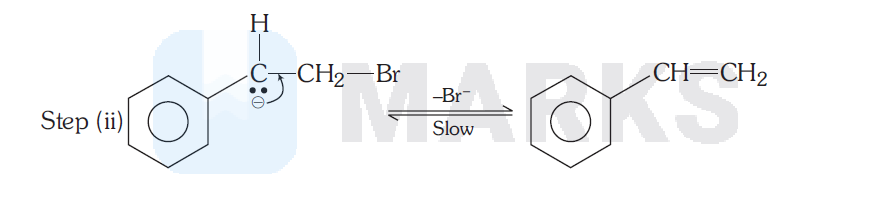
In the step (II), a slow unimolecular elimination occurs in the conjugate base of the reactant and hence this mechanism is called $E_1 C B$ or carbanion mechanism. Since step (I) must be reversible, if ethanol containing $\mathrm{C}_2 \mathrm{H}_5 \mathrm{OD}$ is used as solvent, it would be expected that the original bromide would incorporate deuterium (D).
$$
\text { (D) Match with (P) and (S) }
$$
Step I. $\mathrm{PhCH}_2-\mathrm{CH}_2-\mathrm{Br} \stackrel{\text { Slow }}{\longrightarrow} \mathrm{PhCH}_2-\stackrel{+}{\mathrm{C}} \mathrm{H}_2+\mathrm{Br}^{-}$
Step II. $\mathrm{PhCH}_2-\stackrel{+}{\mathrm{C}_2} \mathrm{H}_2 \stackrel{\text { Fast }}{\longrightarrow} \mathrm{Ph}-\mathrm{CH}=\mathrm{CH}_2+\mathrm{H}^{-}$
Rate $\propto\left[\mathrm{PhCH}_2-\mathrm{CH}_2-\mathrm{Br}\right]$
Similarly
Step II. $\mathrm{PhCD}_2 \stackrel{+}{\mathrm{C}_2} \mathrm{H}_2 \stackrel{\text { Fast }}{\longrightarrow} \mathrm{PhCD}=\mathrm{CHD}+\mathrm{H}^{+}$
Rate $\propto\left[\mathrm{PhCD}_2-\mathrm{CH}_2 \mathrm{Br}\right]$
Hence, $E_1$ reaction and first order kinetics.
\text { (A) Match with (Q) }
$$
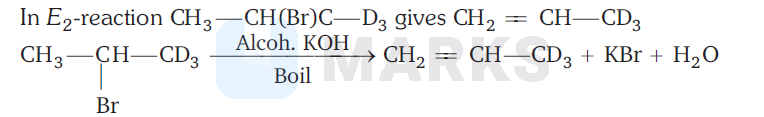
The formation of $\mathrm{CH}_2=\mathrm{CH}-\mathrm{CD}_3$ can be explained on the basis of the fact that $\mathrm{C}-\mathrm{D}$ bond is much stronger than $\mathrm{C}-\mathrm{H}$ bond.
(B) match with (Q)
Reactivity of $\mathrm{PhCHBrCH}_3$ is greater than
$\mathrm{Ph} \mathrm{CHBrCD}_3$ because $\mathrm{C}-\mathrm{D}$ bond is more stronger than $\mathrm{C}-\mathrm{H}$ bond.
$$
\text { (C) match with (R) and (S) }
$$


In the step (II), a slow unimolecular elimination occurs in the conjugate base of the reactant and hence this mechanism is called $E_1 C B$ or carbanion mechanism. Since step (I) must be reversible, if ethanol containing $\mathrm{C}_2 \mathrm{H}_5 \mathrm{OD}$ is used as solvent, it would be expected that the original bromide would incorporate deuterium (D).
$$
\text { (D) Match with (P) and (S) }
$$
Step I. $\mathrm{PhCH}_2-\mathrm{CH}_2-\mathrm{Br} \stackrel{\text { Slow }}{\longrightarrow} \mathrm{PhCH}_2-\stackrel{+}{\mathrm{C}} \mathrm{H}_2+\mathrm{Br}^{-}$
Step II. $\mathrm{PhCH}_2-\stackrel{+}{\mathrm{C}_2} \mathrm{H}_2 \stackrel{\text { Fast }}{\longrightarrow} \mathrm{Ph}-\mathrm{CH}=\mathrm{CH}_2+\mathrm{H}^{-}$
Rate $\propto\left[\mathrm{PhCH}_2-\mathrm{CH}_2-\mathrm{Br}\right]$
Similarly
Step II. $\mathrm{PhCD}_2 \stackrel{+}{\mathrm{C}_2} \mathrm{H}_2 \stackrel{\text { Fast }}{\longrightarrow} \mathrm{PhCD}=\mathrm{CHD}+\mathrm{H}^{+}$
Rate $\propto\left[\mathrm{PhCD}_2-\mathrm{CH}_2 \mathrm{Br}\right]$
Hence, $E_1$ reaction and first order kinetics.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.