Search any question & find its solution
Question:
Answered & Verified by Expert
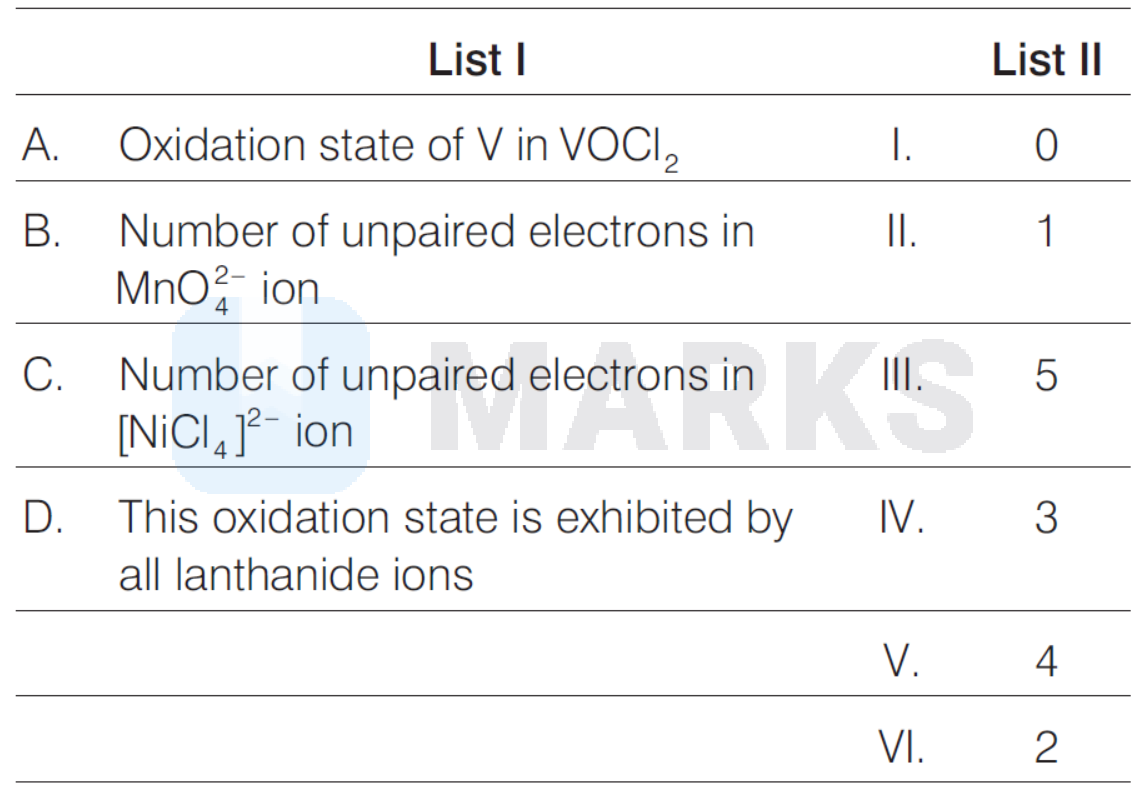
Match the following:
The correct answer is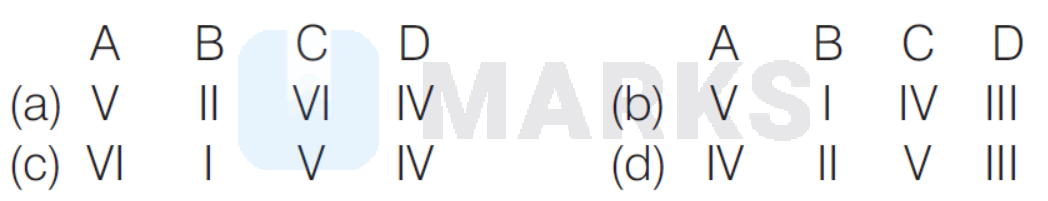
Options:

The correct answer is
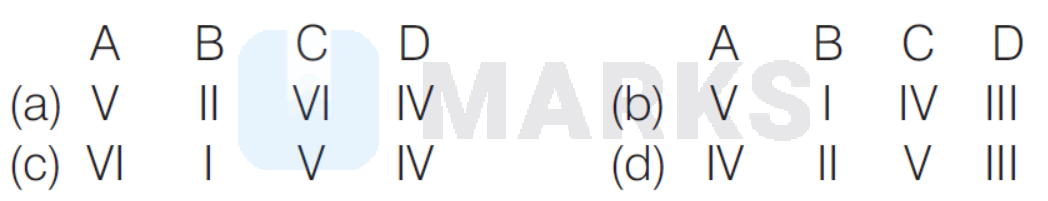
Solution:
2791 Upvotes
Verified Answer
The correct answer is:
(a)
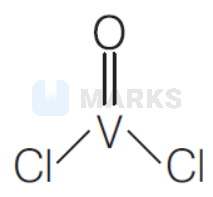
(A) Oxidation state of $\mathrm{VOCl}_2$ is +4 .
Oxidation state of $\mathrm{V}=x$
$$
\begin{array}{rlrl}
& & x-2-2 & =0 \\
\Rightarrow & x & =+4
\end{array}
$$
Thus, (A) is correct match with (V).
(B)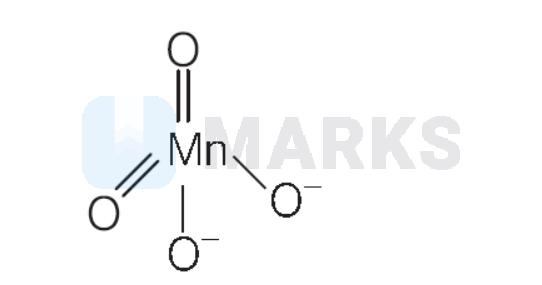
Oxidation state of $\mathrm{Mn}=x$
$$
\begin{gathered}
x-8=-2 \\
x=+6 \\
\mathrm{Mn}^{+6} \longrightarrow[\mathrm{Ar}] 3 d^1 4 s^0
\end{gathered}
$$
$\mathrm{Mn}^{+6}$ contains one unpaired electron in $d$-subshell. Hence, (B) is correct match with (II).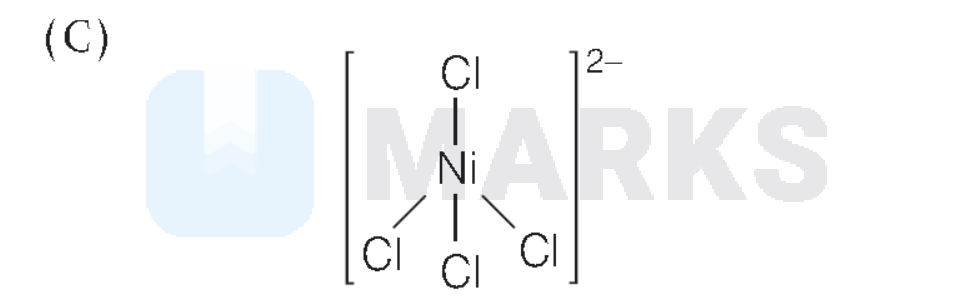
Oxidation state of $\mathrm{Ni}=x$
$$
\begin{aligned}
x-4 & =-2 \\
x & =+2
\end{aligned}
$$
$$
\mathrm{Ni}^{2+} \rightarrow[\mathrm{Ar}] 3 d^8 4 s^0
$$
$\mathrm{Ni}^{2+}$ has two unpaired electron in $d$-subshell. Thus, (C) is correct match with (VI).
(D) Lanthanide ions exhibited by oxidation state equal to +3 . Hence, (D) is correct match with (IV).
Oxidation state of $\mathrm{V}=x$
$$
\begin{array}{rlrl}
& & x-2-2 & =0 \\
\Rightarrow & x & =+4
\end{array}
$$

Thus, (A) is correct match with (V).
(B)

Oxidation state of $\mathrm{Mn}=x$
$$
\begin{gathered}
x-8=-2 \\
x=+6 \\
\mathrm{Mn}^{+6} \longrightarrow[\mathrm{Ar}] 3 d^1 4 s^0
\end{gathered}
$$
$\mathrm{Mn}^{+6}$ contains one unpaired electron in $d$-subshell. Hence, (B) is correct match with (II).

Oxidation state of $\mathrm{Ni}=x$
$$
\begin{aligned}
x-4 & =-2 \\
x & =+2
\end{aligned}
$$
$$
\mathrm{Ni}^{2+} \rightarrow[\mathrm{Ar}] 3 d^8 4 s^0
$$
$\mathrm{Ni}^{2+}$ has two unpaired electron in $d$-subshell. Thus, (C) is correct match with (VI).
(D) Lanthanide ions exhibited by oxidation state equal to +3 . Hence, (D) is correct match with (IV).
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.