Search any question & find its solution
Question:
Answered & Verified by Expert
\( \mathrm{A} \) (Light pink colour complex) \( \frac{\mathrm{Pb}_{3} \mathrm{O}_{4} / \mathrm{dil} . \mathrm{HNO}_{3}}{\Delta} \mathrm{HMnO}_{4} \stackrel{\mathrm{H}_{2} \mathrm{~S} / \mathrm{H}^{+}}{\longrightarrow} \mathrm{A} \) (Light pink colour complex)
Calculate the sum total of CFSE value in light pink colour complex for both sets of orbitals as compared to the average
energy of \( \mathrm{d} \)-orbital.
Calculate the sum total of CFSE value in light pink colour complex for both sets of orbitals as compared to the average
energy of \( \mathrm{d} \)-orbital.
Solution:
1182 Upvotes
Verified Answer
The correct answer is:
0
,
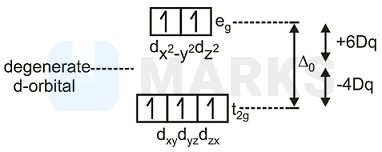
of the pink colour complex
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.